ABSTRACT
Even though plants containing the alkaloid berberine have been used in Ayurvedic and Chinese medicine for over 2500 years, berberine’s potential cardiovascular and metabolic effects have been studied only in the recent past. The objective of this review is to summarize the effects and possible mechanisms of action of berberine when applied to various aspects of the cardiovascular system. Evidence is presented from experimental studies, clinical trials, and meta-analyses accessed via PubMed. Further research is needed, particularly clinical trials, but a growing body of evidence suggests an important role for berberine in the treatment of dyslipidemia, type 2 diabetes, and metabolic syndrome.
INTRODUCTION
Berberine is an intensely yellow-colored, bitter isoquinolone alkaloid that is found in several plants, including Coptis chinensis (Chinese goldthread), Coptis trifolia (American goldthread), Mahonia aquifolium (Oregon grape), Berberis vulgaris (common barberry), and Hydrastis canadensis (goldenseal).1 Berberine has been studied extensively for its antimicrobial, immunostimulatory, anticonvulsant, sedative, hypotensive, uterotonic, choleretic, anticancer,2 antihelminthic,3 and carminative activities.4 More recently, its impact on carbohydrate and lipid metabolism, endothelial function, and cardiotonicity have made it one of the most researched alkaloids of the last decade.
CHEMICAL CHARACTERISTICS AND PHARMACOKINETICS
Berberine is classified as an isoquinoline alkaloid (2,3-methylenedioxy-9,10-di-methoxyprotoberberine chloride [C20H18NO4+]) and has a molecular mass of 336.36122 g/mol.5Figure 1 illustrates its chemical structure. The oral bioavailability of berberine is low (<5%) because of both poor solubility and the presence of P-glycoprotein in the human intestines, which expels the alkaloid from the mucosal lumen.6 As a consequence, significantly large doses of berberine are needed for a therapeutic effect. Berberine is converted in the gut to dihydroberberine. This biological derivative is five times more absorbable.7
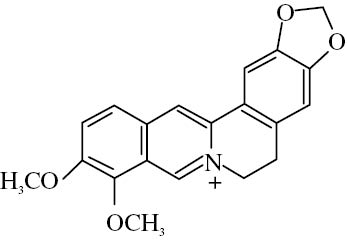
Figure 1:
Chemical structure of berberine.
VASCULAR ENDOTHELIUM AND THE HEART
Endothelial dysfunction contributes to most cardiovascular problems, including plaque initiation and progression, hypertension, coronary artery disease, chronic heart failure, and peripheral artery disease.8 Alteration of endothelial homeostasis is due primarily to proinflammatory cytokines and reduced adiponectin secretion, which are key events in obesity, insulin resistance, and hyperglycemia. These conditions are associated with altered gene expression and cell signaling in the vascular endothelium, thereby affecting release of endothelium-derived factors, activation of NADPH oxidase, uncoupling of endothelial nitric oxide synthase (eNOS), and the expression of endothelin-1. The end result is an imbalance between the production of vasodilator and vasoconstrictor mediators, and the induction of adhesion molecules.9Figures 2 and 3 illustrate how berberine acts at various levels to restore endothelial homeostasis.
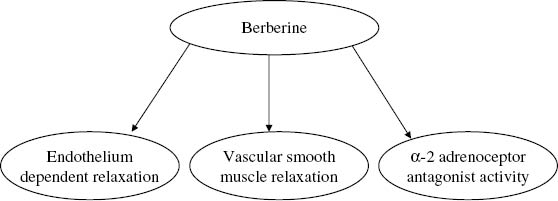
Figure 2:
Effects of berberine on endothelial homeostasis.
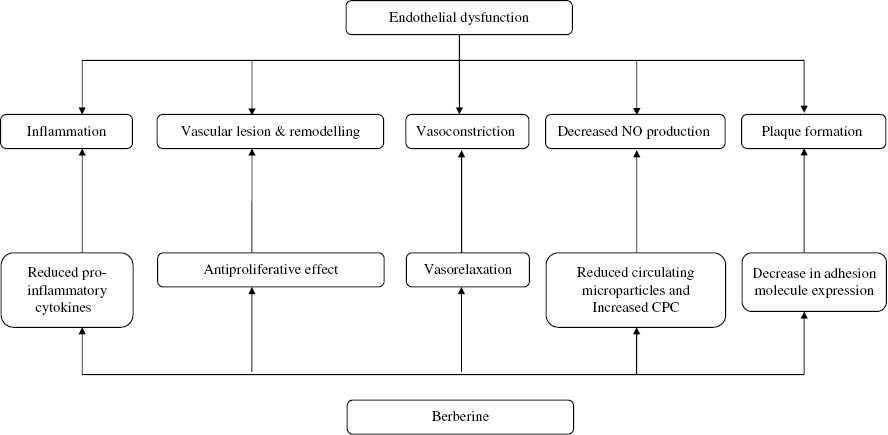
Figure 3:
Effects of berberine on endothelium and heart. (Reproduced from Affuso F, MercurioV, Fazio V, Fazio S. Cardiovascular and metabolic effects of berberine. World J Cardiol. 2010;2:71–7, with permission.10)
The fundamental mechanism by which berberine appears to have such an extensive impact is likely to involve adenosine monophosphate–activated protein kinase (AMPK). Endothelial AMPK plays a major role in restoring vascular homeostasis by mediating eNOS activation11; modulating cellular energy12; preventing apoptosis13; and regulating inflammation, angiogenesis, and perfusion.14,15 Berberine inhibits the intracellular accumulation of reactive oxygen species, cellular apoptosis, and inflammation that characterize vascular injury, and these are mostly triggered by hyperglycemia.10 Berberine thus has antiproliferative and vasoprotective actions.
In vitro studies have demonstrated that berberine acts on both endothelial and underlying vascular smooth muscle cells (VSMCs) to induce vasodilation.16 This action is mediated by eNOS, which leads to nitric oxide (NO) production via the AMPK cascade. Downregulation of eNOS is associated with metabolic conditions that lead to altered insulin signaling, such as diabetes and hypercholesterolemia.17,18 Berberine, when combined with policosanols and red yeast rice, was shown to have a positive effect on flow-mediated vasodilation in a double-blind, placebo-controlled trial of subjects with hypercholesterolemia.19
Berberine reduces the number of adherent monocytes on endothelial cells, which is an early indicator of atherosclerosis. Berberine also suppresses proinflammatory cytokines, which are induced by hyperglycemia and involved in the development of atherosclerotic plaques.20
BLOOD PRESSURE
Berberine was shown to reduce blood pressure by competitively inhibiting VSMC α1 receptors, thereby blocking the release of the enzyme adenylyl cyclase. This resulted in vasodilation and augmented acetylcholine activity.21 Reduction in blood pressure and vasodilation are caused by both vascular smooth muscle relaxation and endothelium-dependent relaxation. Berberine induced endothelium-dependent relaxation by increasing endothelial NO release.22 At a dose of 5–10 mg/kg, berberine improved cardiac contractility, inhibited myocardial fibrosis, and reduced cardiac atrophy in a rat hypertension model.23 Berberine was also shown to function like a calcium channel blocker to reduce high blood pressure and vascular hardening.24
GLUCOSE REGULATION
AMPK plays a key role in regulating cellular and whole-body energy homeostasis. Berberine’s effects on glucose metabolism are due in part to its activation of AMPK.25 These effects are summarized in Figure 4. In a mouse model of diabetes, berberine was shown to modulate the expression of genes that promote catabolism of high-energy intermediates and to promote glucose uptake through a mechanism distinct from insulin. Insulin increases cellular glucose uptake by promoting glucose transporter type 4 (GLUT4) expression on the cell surface through the activation of phosphatidylinositol 3-kinase. By contrast, berberine appeared to induce glucose transport by enhancing GLUT1 gene expression.26 These effects are mediated by the activation of AMPK, which coordinates both short- and long-term metabolic changes, leading to an improvement in energy production and a reduction in energy storage. Specifically, AMPK activation leads to an increase in the uptake of glucose from the blood to target organs.
Other studies have highlighted complex I, the largest enzyme complex of the mitochondrial respiratory chain, as a major target for berberine.27 Berberine activates AMPK and induces glycolysis, resulting in lowered insulin resistance and decreased oxygen respiration.28 Another mechanism underlying the action of berberine on insulin sensitivity is its ability to increase insulin receptor (InsR) expression in a dose- and time-dependent manner, thereby promoting cellular glucose uptake in the presence of insulin. Berberine induces InsR gene expression via transcriptional regulation through protein kinase C (PKC). In a mouse model of type 2 diabetes, berberine lowered fasting blood glucose and fasting serum insulin. It increased insulin sensitivity and elevated InsR mRNA, as well as PKC activity in the liver. As expected, berberine did not lower blood glucose in type 1 diabetic mice, because of insulin deficiency.29 These results were consistent with those of a double-blind, placebo-controlled trial in which berberine administration lowered fasting and postprandial plasma glucose levels, with a slight reduction in postprandial insulin and body weight in patients with type 2 diabetes.30
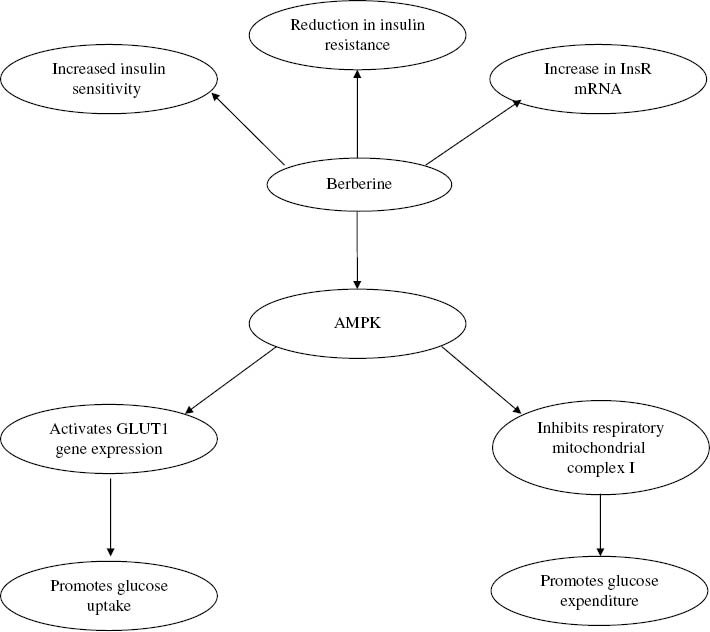
Figure 4:
Effects of berberine on glucose metabolism.
Berberine was also shown to lower fasting blood glucose, hemoglobin A1c, and insulin levels in patients with type 2 diabetes at levels similar to metformin and rosiglitazone, confirming the upregulation of InsR.31 Berberine has an additional advantage over thiazolidinediones (TZDs) in that it inhibits the differentiation of preadipocytes, reduces accumulation of lipid droplets, and lowers triglyceride levels.32–34 Unlike TZDs, which may lead to weight gain, berberine may be better suited for insulin-resistant and obese patients with diabetes.
BLOOD LIPIDS
Berberine has been shown to reduce plasma cholesterol and triglyceride (TG) levels in both human and animal trials. The mechanism of action appears to be downregulation of low-density lipoprotein cholesterol (LDL-C) by upregulation of LDL receptor (LDLR) expression. This mechanism is distinct from how statins work but comparable in effect.35,36 In addition to upregulating LDLR expression, berberine reduces cholesterol levels by inhibiting cholesterol absorption and promoting its excretion (Figure 5). Berberine treatment (50–150 mg/kg) in atherogenic rats reduced total cholesterol (TC) by 29%–33% and non–high-density lipoprotein (non-HDL) by 31%–41%. It also reduced the absorption rate of fractional dietary cholesterol by 40%–51%.37 These findings point to the connection between plasma TC or non-HDL levels and cholesterol absorption rates, owing to the decrease of enterocyte cholesterol uptake and secretion.
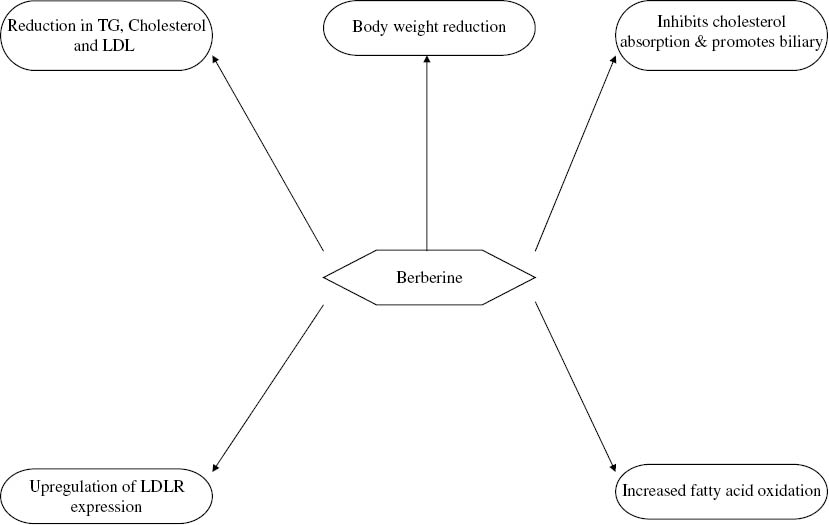
Figure 5:
Antihyperlipidemic actions of berberine.
Berberine’s lipid-lowering effect is also likely due to the promotion of cholesterol excretion from the liver into the bile. This was demonstrated in a study of hyperlipidemic hamsters treated with either 50 or 100 mg/kg berberine. A gradual decrease in liver cholesterol levels and an increase in bile cholesterol levels was observed at both doses.38 Another experiment using hyperlipidemic hamsters demonstrated that berberine and plant stanols worked synergistically to inhibit cholesterol absorption more effectively than berberine or plant stanols alone.39
The hypolipidemic action of berberine appears to be enhanced when it is combined with policosanols and red yeast rice.40 The efficacy of this same combination was confirmed in a double-blind, placebo-controlled trial in which a significant reduction of total LDL-C and TG was seen in participants with hypercholesterolemia.19 It is important to note that, compared with simvastatin therapy, which has been shown to maximally reduce LDL-C to 60%, the effect of berberine alone appears to be moderate.41
CONGESTIVE HEART FAILURE
In a double-blind, placebo-controlled study, participants with congestive heart failure (CHF) who received berberine at a dose of 1.2–2.0 g/day showed significant improvement in a 6-minute walking distance, left ventricular ejection fraction, ventricular premature complexes, and nonsustained ventricular tachycardia at the end of 8 weeks of treatment and at 24-month follow-up. No side effects were reported. In addition, total mortality in the berberine treatment group was 8.8% compared with 16.4% for control subjects.42 Acute infusion of berberine at a rate of 0.2 mg/kg/min was found to improve hemodynamics in patients with heart failure who were refractory to digitalis and diuretics. Specifically, it reduced systemic and pulmonary vascular resistance and left ventricular end-diastolic pressure.43
In a rat model of cardiac hypertrophy, animals administered berberine at a dose of 10 mg/kg body weight for 8 weeks beginning 4 weeks after aortic banding had statistically significant reductions in whole-heart and left ventricle size, as well as decreased left ventricular end-diastolic pressure, compared with controls.44 In a dog model of cardiac ischemia, intravenous administration of berberine (1 mg/kg within 3 minutes of left ventricular failure) followed by a constant infusion (0.2 mg/kg/min for 30 minutes) over 10 days improved cardiac output. Berberine also decreased left ventricular end-diastolic pressure, diastolic blood pressure, and systemic vascular resistance, but it did not affect heart rate. This suggests that berberine may be able to improve impaired left ventricular function by exerting positive ionotropic effects and mild systemic vasodilation.45
INTERACTIONS, ADVERSE EFFECTS, AND SAFETY ISSUES
Berberine is generally well tolerated in therapeutic doses, with rare adverse events reported. High doses of berberine may cause arterial hypotension, dyspnea, flulike symptoms, gastrointestinal discomfort, constipation, cardiac damage, and gastric lesions.46 An experimental study using rats found that daily intraperitoneal administration of berberine in doses of 10 and 20 µg/g for 1 week displaced bilirubin from albumin.47 Any berberine-containing herb therefore should be avoided in large doses in jaundiced infants and pregnant women due to the risk of bilirubin-induced brain damage. Berberine can displace warfarin, thiopental, and tolbutamide from their binding sites, increasing their levels in the plasma.48 Allergic reactions have been reported after intravenous administration of berberine.49
CONCLUSION
The cardiovascular effects of berberine appear to be mediated through the AMPK cascade, which is the main pathway implicated in cardiovascular and metabolic disorders. By restoring endothelial homeostasis and reducing blood pressure, berberine acts as a potent antihypertensive and cardioprotective agent. Many studies indicate that berberine can reduce blood glucose, hemoglobin A1c, and insulin levels in patients with type 2 diabetes. It may also improve insulin resistance and reduce body weight. As a result, berberine is a promising addition to the toolbox of therapies for treating type 2 diabetes and metabolic syndrome.
Berberine’s safety profile and the favorable results from combination therapy support its use in patients with mild hyperlipidemia, as well as for patients who do not tolerate statins or do not achieve therapeutic goals. Clinical trials of human subjects with type 2 diabetes, metabolic syndrome, or CHF suggest that a safe and effective dosage range for berberine is between 0.5 and 1.5 g/day, usually in divided doses.30,50,51 In clinical practice, this dosage range may vary if the whole berberine-containing plant is used and with different extracts or concentrations. The actions of berberine are summarized in Figure 6.
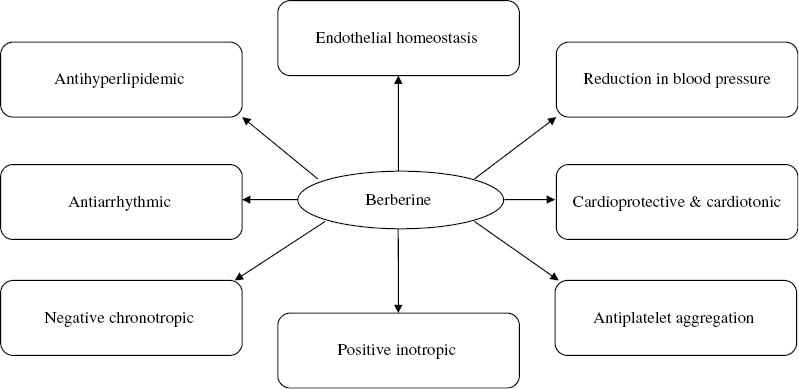
Figure 6:
Summary of the actions of berberine.
The authors wish to thank Elizabeth Sutherland, ND for her invaluable contribution to the development of this paper.
The authors declare they have no competing interests.
- Kamath S,
Skeels M,
Pai A.
Significant differences in alkaloid content of Coptis chinensis (Huanglian), from its related American species.
- Gan RY.
Bioactivities of berberine: an update.
- Kumar A, Ekavali,
Chopra K,
et al.
Current knowledge and pharmacological profile of berberine: an update.
- Meneton P,
Jeunemaitre X,
de Wardener HE,
MacGregor GA.
Links between dietary salt intake, renal salt handling, blood pressure, and cardiovascular diseases.
- Xia LM,
Luo MH.
Study progress of berberine for treating cardiovascular disease.
- Pan GY,
Wang GJ,
Liu XD,
et al.
The involvement of p-glycoprotein in berberine absorption.
- Feng R,
Shou JW,
Zhao ZX, et al. Transforming berberine into its intestine-absorbable form by the gut microbiota.
- Endemann DH,
Schiffrin EL.
Endothelial dysfunction.
- Rask-Madsen C,
King GL.
Mechanisms of disease: endothelial dysfunction in insulin resistance and diabetes.
- Affuso F, MercurioV, Fazio V,
Fazio S.
Cardiovascular and metabolic effects of berberine.
- Zhang Y,
Lee TS,
Kolb EM,
et al.
AMP-activated protein kinase is involved in endothelial NO synthase activation in response to shear stress.
- Dagher Z,
Ruderman N,
Tornheim K,
Ido Y.
The effect of AMP-activated protein kinase and its activator AICAR on the metabolism of human umbilical vein endothelial cells.
- Ido Y,
Carling D,
Ruderman N.
Hyperglycemia-induced apoptosis in human umbilical vein endothelial cells: inhibition by the AMP-activated protein kinase activation.
- Nagata D,
Mogi M,
Walsh K.
AMP-activated protein kinase (AMPK) signaling in endothelial cells is essential for angiogenesis in response to hypoxic stress.
- Ouchi N,
Shibata R,
Walsh K.
AMP-activated protein kinase signaling stimulates VEGF expression and angiogenesis in skeletal muscle.
- Hui KK,
Yu JL,
Chan WF,
et al.
Interaction of berberine with human platelet alpha 2 adrenoceptors.
- Henry PD,
Cabello OA,
Chen CH.
Hypercholesterolemia and endothelial dysfunction.
- Montagnani M,
Ravichandran LV,
Chen H,
et al.
Insulin receptor substrate-1 and phosphoinositide-dependent kinase-1 are required for insulin-stimulated production of nitric oxide in endothelial cells.
- Affuso F,
Ruvolo A,
Micillo F,
et al.
Effects of a nutraceutical combination (berberine, red yeast rice and policosanols) on lipid levels and endothelial function randomized, double-blind, placebo-controlled study.
- Wang Y,
Huang Y,
Lam KS,
et al.
Berberine prevents hyperglycemia induced endothelial injury and enhances vasodilatation via adenosine monophosphate-activated protein kinase and endothelial nitric oxide synthase.
- Zhang LS.
Clinical usage of berberine.
- Ko WH,
Yao XQ,
Lau CW,
et al.
Vasorelaxant and antiproliferative effects of berberine.
- Zhao HP,
Hong Y,
Xie JD,
et al.
Effect of berberine on left ventricular remodeling in renovascular hypertensive rats [in Chinese].
- Wang J,
Guo T,
Peng QS,
et al.
Berberine via suppression of transient receptor potential vanilloid 4 channel improves vascular stiffness in mice.
- Lee YS,
Kim WS,
Kim KH,
et al.
Berberine, a natural plant product, activates AMP-activated protein kinase with beneficial metabolic effects in diabetic and insulin-resistant states.
- Kim SH,
Shin EJ,
Kim ED,
et al.
Berberine activates GLUT1-mediated glucose uptake in 3T3-L1 adipocytes.
- Turner N,
Li JY,
Gosby A,
et al.
Berberine and its more biologically available derivative, dihydroberberine, inhibit mitochondrial respiratory complex I: a mechanism for the action of berberine to activate AMP-activated protein kinase and improve insulin action.
- Yin J,
Gao Z,
Liu D,
et al.
Berberine improves glucose metabolism through induction of glycolysis.
- Kong WJ,
Zhang H,
Song DQ,
et al.
Berberine reduces insulin resistance through protein kinase C-dependent up-regulation of insulin receptor expression.
- Zhang Y,
Li X,
Zou D,
et al.
Treatment of type 2 diabetes and dyslipidemia with the natural plant alkaloid berberine.
- Zhang H,
Wei J,
Xue R,
et al.
Berberine lowers blood glucose in type 2 diabetes mellitus patients through increasing insulin receptor expression.
- Liu Y,
Lou SY,
He YM.
Effects of berberine on cell proliferation, peroxisome proliferation activated receptor gamma, CAAT/enhancer binding protein mRNA and protein expression in 3T3-L1 pre-adipocytes [in Chinese].
- Zhou JY,
Zhou SW,
Zhang KB,
et al.
Chronic effects of berberine on blood, liver glucolipid metabolism and liver PPARs expression in diabetic hyperlipidemic rats.
- Huang C,
Zhang Y,
Gong Z,
et al.
Berberine inhibits 3T3-L1 adipocyte differentiation through the PPARgamma pathway.
- Kong W,
Wei J,
Abidi P,
et al.
Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins.
- Li H,
Chen W,
Zhou Y,
et al.
Identification of mRNA binding proteins that regulate the stability of LDL receptor mRNA through AU-rich elements.
- Wang Y,
Yi X,
Ghanam K,
et al.
Berberine decreases cholesterol levels in rats through multiple mechanisms, including inhibition of cholesterol absorption.
- Li XY,
Zhao ZX,
Huang M,
et al.
Effect of berberine on promoting the excretion of cholesterol in high-fat diet-induced hyperlipidemic hamsters.
- Wang Y,
Jia X,
Ghanam K,
et al.
Berberine and plant stanols synergistically inhibit cholesterol absorption in hamsters.
- Cicero AF,
Rovati LC,
Setnikar I.
Eulipidemic effects of berberine administered alone or in combination with other natural cholesterol-lowering agents: a single-blind clinical investigation.
- Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial.
- Zeng XH,
Zeng XJ,
Li YY.
Efficacy and safety of berberine for congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy.
- Marin-Neto JA,
Maciel BC,
Secches AL,
et al.
Cardiovascular effects of berberine in patients with severe congestive heart failure.
- Hong Y,
Hui SS,
Chan BT,
Hou J.
Effect of berberine on regression of pressure- overload induces cardiac hypertrophy in rats.
- Huang WM,
Yan H,
Jin JM,
et al.
Beneficial effects of berberine on hemodynamics during acute ischemic left ventricular failure in dogs.
- Imanshahidi M,
Hosseinzadeh H.
Pharmacological and therapeutic effects of Berberis vulgaris and its active constituent, berberine.
- Chan E.
Displacement of bilirubin from albumin by berberine.
- Tan YZ,
Wu AC,
Tan BY,
et al.
Study on the interactions of berberine displace other drug from their plasma proteins binding sites.
- Wang YS.
- Zilaee M,
Kermany T,
Tavalaee S, et al.
Barberry treatment reduces serum anti-heat shock protein 27 and 60 antibody titers and high- sensitivity C-reactive protein in patients with metabolic syndrome: a double blind, randomized, placebo controlled trial.
- Yarnell E.
Recent clinical advances with berberine.