ABSTRACT
Background: Iodine is a vital micronutrient essential for thyroid hormone synthesis and neurological development in utero and in the early postpartum period. Maternal iodine deficiency during pregnancy can result in infant congenital hypothyroidism. Congenital hypothyroidism is the most prevalent preventable cause of mental retardation in the world. Some proponents of supplementation believe that products containing both iodine (I2) and iodide (I−) are therapeutically superior to iodide-only formulations. As a step toward evaluating this claim, we tested three commercially available products that list both iodine and iodide on the label, to determine their content.
Design: Two powdered and one aqueous solution were tested using ion chromatography, X-ray spectrometry, ion coupled mass spectrometry, and the iodine-starch test to more accurately determine their composition.
Results: We found that when each of the dry supplements were dissolved in a slightly basic solution, only the I− form was then present.
Conclusion: Our findings suggest that dry products made with only iodide are likely to be substantially equivalent to dry products made with both forms, at least by the time they are digested and absorbed.
INTRODUCTION
Iodine is a vital micronutrient that is essential for thyroid hormone synthesis and for neurological development in the fetus and in the early postpartum period. Maternal iodine deficiency during pregnancy and lactation can cause infant congenital hypothyroidism.1 Congenital hypothyroidism is the leading preventable cause of severe developmental disability (such as low IQ) in the world.2 Iodized salt was introduced in the United States in the 1920s to address the alarming rates of iodine deficiency disease, including goiter and hypothyroidism, that were prevalent in certain regions. In 1922, the incidence of goiter in some parts of Michigan was as high as 64%.3 In North America, 90% of households consume iodized salt, though iodine deficiency is still prevalent in some populations.4
Iodine concentration is highest in the thyroid gland (70%–80%). The rest is stored in the mammary glands, salivary glands, gastric mucosa, and cervix,5 with some distribution in bone, skeletal muscle, skin, kidneys, liver, lung, spleen, and adipose tissue.6 Median urinary iodine levels are used to determine iodine sufficiency across populations. The World Health Organization defines mild iodine insufficiency as urine iodine concentrations below 150 µg/L for pregnant and lactating women and below 100 µg/L for other adults and children.7 Moderate iodine insufficiency is below 50 µg/L. The National Health and Nutrition Examination Survey (NHANES), a large US-based population study, determined that urine iodine concentrations sharply decreased in the United States from 320±6 µg/L in NHANES I (1971–1974) to 145±3 µg/L in NHANES II ((1988–1994). In subsequent years, concentrations seemed to stabilize at 168 µg/L according to NHANES 2001–2002 data and at 144 µg/L based on NHANES 2009–2010 data.8 A compilation of Japanese studies found 24-hour urine iodine levels as high as 23,300 µg/L in 1965 and 3300 µg/L in 1994.9 The intake of iodine from seaweed in the traditional Japanese diet is one of the highest in the world, which may contribute to the lower incidence of fibrocystic breast disease and breast cancer observed in Japan.10
FORMS OF IODINE
Elemental diatomic iodine (I2) and ionic monoatomic iodide (I−) are two forms of iodine. Elemental diatomic iodine rarely occurs in nature. Almost all forms found in nature occur as iodide salts, most commonly as inorganic sodium and potassium salts (iodides and iodates). Iodate can be reduced to I2, which is a reactive intermediate product that can be caustic to skin and mucus membranes.11,12 Molecular iodine (I2) is absorbed through the intestinal tract by facilitated diffusion, whereas iodide (I−) is absorbed via a transport protein in the gastric mucosa called the sodium-iodide symporter (NIS).13 NIS sequesters iodide and can concentrate iodine levels in tissues up to 20–40 times greater than that in serum. Tissues containing NIS include the thyroid, salivary glands, mammary glands, gastric mucosa,13 ovaries and fallopian tubes,14 testes,15 and placenta.16 I2 is readily converted to I− in the gut and rapidly absorbed into the bloodstream.17
IODINE PHYSIOLOGY
Iodine uptake and use in the thyroid and mammary glands are essential for the health of the developing fetus. Animal studies have shown that during lactation, iodine is concentrated in the mammary glands to a greater degree than in the thyroid gland.18 Increased expression of NIS on lactating breast cells means that iodine is secreted into human breast milk at a concentration gradient 20–50 times that of plasma.2 This evolutionary mechanism is necessary for neonatal thyroid function and consequent healthy neural development.19 Mammary tissue also produces two separate deiodinase enzymes: deiodinase type 1 and deiodinase type 2, which can convert thyroxine (T4) to triiodothyronine (T3). Deiodinases control the amount of free iodine present in breast tissue – higher levels during puberty, pregnancy, and lactation and lower levels in nonpregnant or postpartum periods.20 The thyroid and mammary tissue oxidize iodide to iodine during the process of organification that produces iodoproteins.21,22 This process requires the presence of H2O2 as an oxidizing agent catalyzed by thyroid peroxidase (TPO) in the thyroid and by lactoperoxidases (LPO) in the breast (Figure 1).
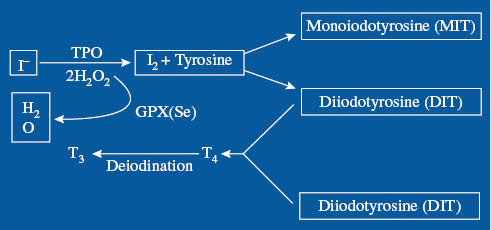
Figure 1:
Antioxidant functions of iodine.23
Iodine (I2) is catalyzed by thyroid peroxidase using H2O2. H2O2 used in this reaction decreases the amount of H2O2 that would otherwise be available for damaging oxidation reactions. Selenium containing GPX removes H2O2 from the tissues, also decreasing oxidative damage. GPX, glutathione peroxidase; T3, triiodothyronine; T4, tetraiodothyronine; TPO, thyroperoxidase. Reproduced with Permission from Patrick L. Iodine: Deficiency and therapeutic considerations. Alternative Medicine Review 2008;13:116–127.
SUPPLEMENTATION: IODINE, IODIDE, OR BOTH?
Some proponents of dietary supplementation believe that a combination of iodine and iodide may be superior to iodide alone, because each form may have different therapeutic effects. A few animal and in vitro studies have suggested a difference,24–26 but human studies have not.9 Animal studies that showed different effects of iodine and iodide might have done so as a consequence of flaws in study design. For example, in a study where rats were given estrogen to induce dysplasia and perchlorate to induce iodine deficiency, followed by either iodine (I2) or iodide (I−), I2 was found to be more effective than I− in reversing atypical ductal hyperplasia in mammary gland tissue.22 This result could, however, be explained in part because perchlorate, which is a competitive inhibitor of iodide uptake by the NIS,27 was administered to induce iodine deficiency, and iodine (I2) is internalized more rapidly and effectively than iodide in the absence of NIS.18 Closer inspection of the findings of an experimental study that looked at the effects of different combinations of iodine, iodide, or both on mammary gland tumor cells actually showed similar effects for all three as long as the mechanism to convert iodide to iodine was not impaired by, for example, a deficiency in NIS or tissue-specific peroxidases.26
An article that is often cited in support of differential therapeutic claims for iodine and iodide, reported three studies of women with fibrocystic breast disease.28 These studies, however, did not actually compare I2 with I−. The first study compared aqueous I2/I− with iodized casein, a protein-bound form of iodide. For the second study, participants who did not respond to protein-bound iodide were then switched to molecular iodine. The third study was a small controlled trial that compared molecular iodine with placebo. The researchers concluded from these three studies that the molecular form of iodine was more effective in treating fibrocystic breast disease and had a lower side effect profile than iodide, but their results were not statistically significant, and reported side effects appear to be similar for both forms.
Iodine supplementation can be taken therapeutically or to treat deficiencies. In 1829, an aqueous solution of both I2 and I− became available.29 Iodine disinfectant is also an aqueous solution of both I2 and I−, but it is for topical use only. Today many supplements derived from potassium iodide (KI) or a mixture of I2 and I− are available in tablet and encapsulated form for oral consumption. To further investigate the claim that it is better to take both I2 and I−, we decided to test different iodine supplements using ion chromatography (IC), X-ray spectrometry, ion-coupled mass spectrometry, and the iodine-starch test to more accurately determine their composition.
MATERIAL AND METHODS
Three iodine products were tested. Brand 1 was a precisely quantified tablet manufactured from an aqueous solution of iodine and KI. The tablets were formed by spraying the aqueous solution on a colloidal silica excipient until it was absorbed, baking the excipient so that it was dry, pressing it into a tablet, then applying a thin pharmaceutical glaze that consisted of microcrystalline cellulose to eliminate the unpleasant taste of iodine and prevent gastric irritation. On the basis of the amount of iodine and iodide used to make the aqueous solution, each tablet was calculated to be made from 5 mg of iodine (I2) and 7.5 mg of KI.
Brand 2 was a 5% solution of aqueous iodine/iodide manufactured to contain at least 6 mg of KI (4.59 mg iodide) and 3 mg of molecular iodine per 0.06 mL drop. Brand 3 was manufactured to contain 7.5 mg of iodine (crystallized I2); 5 mg of potassium iodide; and several other ingredients, including ascorbic acid, riboflavin, magnesium citrate, zinc citrate, selenium (selenomethionine), copper gluconate, and inositol hexanicotinate. These ingredients were mixed together in a powder form and encapsulated into vegetable cellulose capsules.
Brand 1 was tested for total combined iodine and iodide using X-ray fluorescence. Brand 2 was tested for total combined iodine and iodide using inductively coupled plasma mass spectrometry. All three brands were tested for the presence of iodide (I−) isolates using IC. Brand 1 and brand 2 were also tested for the presence of triiodide using the starch test.
ION CHROMATOGRAPHY
In IC testing, a substance is diluted, dissolved, or extracted into a slightly basic solution. A set of standards is also prepared with concentrations spanning the expected range of the sample solution. A portion of the sample and standard solution are injected into the ion chromatograph. These solutions are run through an anion exchange column that will retain analytes on the basis of their negative charge. The larger the negative charge, the more they will be retained and the longer their retention time will be. The retention time of a particular anion is determined by the standard solution. The anion is detected using a conductivity detector. The detector provides a response factor (RF) for an anion at a particular concentration. Using the RFs from standards of varying concentrations, a linear model is determined and used to analyze the anion concentration of the sample. The detector can detect only components with a charge. Noncharged components will not be detected.
INDUCTIVELY COUPLED PLASMA MASS SPECTROMETRY
The sample is prepared in a microwave nitric acid digester. Once the sample is dissolved, it is introduced into the inductively coupled plasma mass spectrometry (ICP-MS) for analysis. All components of the sample solution are broken down to elemental ions in argon plasma. This plasma is created by alternating radio waves, and electron liberation is assisted by an electrical spark. Once the sample has been broken down into its elemental ions, these ions are then introduced into the mass spectrum analyzer (quadrupole) for analysis based on their mass-to-charge ratio. The ions can be quantified by comparison with a standard solution or event occurrence.
X-RAY FLUORESCENCE
This is a nondestructive elemental surface analysis technique. It does not require any sample preparation except that the surface of the sample be relatively smooth for consistency. The sample is bombarded with short-wavelength X-rays or gamma rays. This energy must be high enough to exceed the binding energy of the inner orbitals of the atom. One or more of the inner orbital electrons will be ejected. Once this occurs, an electron from a higher orbital will fall to fill the inner orbital. In doing so, the energy released is equal to the difference in energy for those specific orbitals. The energy wavelengths can be calculated using Planck’s law and are characteristic of specific elements. The photons are diffracted through a crystal to separate the photons on the basis of the Bragg equation. The photons are then detected and quantified with a scintillation detector.
TRIIODIDE ANALYSIS
Brand 1 and brand 2 were also tested for the presence of both iodine and iodide using triiodide analysis. This analysis is based on the principle that the presence of triiodide in a solution can be confirmed by the addition of amylose, at which point the solution will turn a dark purple. In the case of liquid samples, the sample should be clear and light enough to observe the color change. If this is not the case, the sample should be diluted until the color change can be observed. At this point, the amylose solution can be added and the presence of triiodide determined. In the case of a solid sample, the sample should be ground and dissolved in deionized water. At this point, the amylose solution may be added and the presence of triiodide determined. If no color change is observed, the conclusion is that the sample solution does not contain triiodide. Neither iodine element alone nor iodide ions alone will give the color result.30
RESULTS
BRAND 1: IODINE-SPRAYED SILICA TABLET
The iodide (I−) detected by IC was 14.05 mg (±1.4 mg) and the total iodine (I2+I−) found from X-ray fluorescence was 13.20 mg (±1.1 mg). Each tablet was manufactured to contain at least 12.5 mg of combined iodine/iodide. It appears that all of the iodine in the dissolved tablet solution was in the form of iodide (I−). It may be that the dry tablet did not contain any I2 (the I2 converted to I− during the manufacturing process) or that any I2 that was in the tablet was converted to I− when the tablet was dissolved in the slightly basic solution. Triiodide analysis was negative for the presence of I3−; thus, I2 had been converted to iodide.
BRAND 2: AQUEOUS IODINE PRODUCT
Total iodine found by ICP-MS was 7.64 mg/(0.06 mL)of iodine/iodide. IC detected only 4.35 mg/(0.06 mL) of iodide, which was below the level stated on the product label. Thus, approximately 3.29 mg remained as I2 or I3−. The result of triiodide analysis was immediately positive.
BRAND 3: ENCAPSULATED PRODUCT
IC detected 13 mg of iodide. Brand 3 was manufactured to contain 7.5 mg of iodine (I2) and 5 mg of KI, so it was expected that only 7.5 mg would be present as iodide. However, the results indicated that all of the iodine from the capsule was in the form of iodide once the capsule was dissolved in a slightly basic solution, which was much less than the pH of the small intestines.
DISCUSSION
Iodine (I2) is a purple-black solid at room temperature. When heated, it turns to a gas (sublimation) producing a violet cloud and dissolves readily in many organic solvents, producing violet solutions in most cases and yellow-brown solutions in alcohols. Iodine I2 is very reactive and breaks down into iodide I− very easily. Our results show that when each of the dry supplements (brand 1 and brand 3) was dissolved in a slightly basic solution, only the I− form was then present.
The triiodide analysis was also negative for brand 1 when dissolved in deionized water. The negative triiodide analysis indicates that there was no I3− present. (I3− is generated when I2 and I− are both in solution). Although this product was made with both iodine and iodide, it appears to deliver only iodide. Encapsulating dry I2 crystals directly into supplements can be extremely damaging to metal encapsulating equipment and mixing bowls, and the final product no longer contains I2 when it is dissolved. Consequently, we recommend that dry products contain only iodide.
The aqueous form of iodine/iodide (brand 2) was tested with ion-coupled plasma mass spectrometry and IC. These tests revealed that the expected amounts of iodide and iodine were still present after combination with the mild base. Triiodide must be present for a positive result in triiodide analysis, and this test was immediately positive only for the aqueous iodine supplement (brand 2), confirming the presence of iodide and iodine. Iodine does not dissociate in water, and it stays in its diatomic form. However, in the presence of KI, it dissolves much more readily because of the equilibrium with triiodide (I3−), which is soluble in water because of its negative charge (I−I−I−). The solubility of iodine (I2) is 0.03 g/100 mL at 20°C, and the solubility of KI is 140 g/100 mL at 20°C.
![]()
The dry supplement tablet (brand 1) and capsule (brand 3) that were formulated to contain iodine and iodide were both negative for iodine (I2). This may be because the positively charged ions present in the products reacted with iodine (I2) to form ionic compounds that dissociate in water, thus converting iodine to iodide. These positively charged ions may include silica (Si4+) in the tablet and magnesium (Mg2+), zinc (Zn2+), selenium (Se2+), and copper (Cu+) in the capsule. The ionic compounds that would result would be silica iodide (SiI4), magnesium iodide (MgI2), zinc iodide (ZnI2), seleno-iodide (SeI2), and copper iodide (CuI).
CONCLUSIONS
Although some experimental and animal studies have suggested that iodine and iodide have different therapeutic effects in the body, particularly for mammary tissue, these benefits have not been replicated in human studies. A study in which researchers reported superior benefits for iodine over iodide in women with fibrocystic breast disease actually compared iodine with mixed iodine/iodide, and the findings were not statistically significant. In the experiment we conducted, we found that when dry supplements were dissolved in deionized (distilled) water or a mild base, the resulting solution contained only monoatomic iodide (I−), regardless of how the supplements were initially manufactured. Saliva or taking the dry supplements with water would have the same effect. Thus, any potential benefit from these powdered products comes from iodide. The body easily converts iodide to iodine, provided the conversion mechanisms are not impaired. Dry products made with only iodide are likely to be substantially equivalent to dry products made with both forms. Further human studies are needed to compare the therapeutic efficacy of dry mixed iodide/iodine formulations with iodine-only formulations, but to be accurate, it will first be necessary to demonstrate that a dry form does actually contain both iodine and iodide to begin with or by the time it is digested and absorbed.
The authors wish to thank Elizabeth Sutherland, ND for her invaluable contribution to the development of this paper.
Dr. Hand declares he has no competing interests. Dr. Wilson declares he is a shareholder in Restorative Formulations.
- Haddow JE,
Glenn MD,
Palomaki GE, et al.
Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child.
- Leung AM,
Pearce EN,
Braverman LE.
Iodine nutrition in pregnancy and lactation.
- Markel H.
“When it rains it pours”: endemic goiter, iodized salt, and David Murray Cowie, M.D.
- Hollowell JG,
Staehling NW,
Hannon WH, et al.
Iodine nutrition in the United States: trends and public health implications: iodine excretion data from National Health and Nutrition Examination Surveys I and III (1971–1974 and 1988–1994).
- Costa A,
Testori OB,
Cenderelli C, et al.
Iodine content of human tissues after administration of iodine containing drugs or contrast media.
- Ahad F,
Ganie SA.
Iodine, iodine metabolism and iodine deficiency disorders revisited. Indian J Endocrinol Metab.
2010; 14:13–7. - World Health Organization.
Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programme Managers, 3rd edn.
Geneva (Switzerland): World Health Organization; 2007 (WHO/NHD/01.1) [cited 2017]. Available from: http://apps.who.int/iris/bitstream/10665/43781/1/9789241595827_eng.pdf
Centers for Disease Control and Prevention.
Trace Elements: Iodine.
Available from: https://www.cdc.gov/nutritionreport/99-02/pdf/nr_ch4a.pdf Accessed August 11, 2017.- Zava TT,
Zava DT.
Assessment of Japanese iodine intake based on seaweed consumption in Japan: a literature-based analysis.
doi: https://doi.org/10.1186/1756-6614-4-14. - Cann SA,
van Netten JP,
van Netten C.
Hypothesis: iodine, selenium and the development of breast cancer.
- National Institute of Health, Office of Dietary Supplements.
Iodine: Fact Sheet for Health Professionals.
Available from: https://ods.od.nih.gov/factsheets/Iodine-HealthProfessional/ Accessed August 11, 2017. - Hou X.
Iodine speciation in foodstuff, tissues and environmental samples: iodine species and analytical methods. In:
Preedy VR,
Burrow GN,
Watson RR, eds.
- Portulano C,
Paroder-Belenitsky M,
Carrasco N.
The Na+/I− symporter (NIS): mechanism and medical impact.
doi: https://doi.org/10.1210/er.2012-1036. - Riesco-Eizaguirre G,
Leoni SG,
Mendiola M, et al.
NIS mediates iodide uptake in the female reproductive tract and is a poor prognostic factor in ovarian cancer.
- Russo D,
Scipioni A,
Durante C, et al.
Expression and localization of the sodium/iodide symporter (NIS) in testicular cells.
- Mitchell AM,
Manley SW,
Morris JC, et al.
Sodium iodide symporter (NIS) gene expression in human placenta.
- Paulev PE,
Zubieta-Calleja G.
Thyroid hormones and disorders.
In: - Bretthauer EW.
Milk transfer comparisons of different chemical forms of radioiodine.
- Topper YJ,
Freeman CS.
Multiple hormone interactions in the developmental biology of the mammary gland.
- Aceves C,
Anguiano B,
Delgado G.
Is iodine a gatekeeper of the integrity of the mammary gland? - Taurog A.
Hormone synthesis: thyroid iodine metabolism.
In: Braverman L, Utiger RD, eds. Werner and Ingbar’s The Thyroid. Philadelphia: Lippincott; 1996. pp. 47–81. - Shah NM,
Eskin BA,
Krouse TB,
Sparks CE.
Iodoprotein formation by rat mammary glands during pregnancy and early post-partum period.
- Patrick L.
Iodine: deficiency and therapeutic considerations.
- Thrall KD,
Bull RJ.
Differences in the distribution of iodine and iodide in the Sprague–Dawley rat.
- Thrall KD,
Sauer RL,
Bull RJ.
Evidence for thyroxine formation following iodine administration in Sprague–Dawley rats.
- García-Solís P,
Alfaro Y,
Anguiano B, et al.
Inhibition of induced-MNU mammary carcinogenesis by molecular iodine (I2) but not by iodide (I−) treatment: evidence that I2 prevents cancer promotion.
- Soriano O,
Delgado G,
Anguiano B, et al.
Antineoplastic effect of iodine and iodide in dimethylbenz[a]anthracene-induced mammary tumors: association between lactoperoxidase and estrogen-adduct production.
- Ghent WR,
Eskin BA,
Low DA,
Hill LP.
Iodine replacement in fibrocystic disease of the breast.
- Preedy VR,
Burrow GN,
Watson RR, eds.
- Ophardt CE.
Starch – iodine.
In: Virtual Chembook.
Available from: http://chemistry.elmhurst.edu/vchembook/548starchiodine.html Accessed August 11, 2017.