Abstract
OBJECTIVE:
Currently, the position of the American Thyroid Association is that hypothyroidism causes symptoms of metabolic slowing due to inadequate production of thyroid hormone by the thyroid gland; it is diagnosed by a lab result of a high thyroid stimulating hormone (TSH) level. While hypothyroidism is considered a chronic, incurable condition, the symptoms typically can be controlled by ingestion of thyroxine (T4) for life. Due to this conclusion, medical providers overall adhere to the assumption that people with normal TSH levels can not have thyroid-hormone-responsive symptoms; likewise, thyroid-hormone-responsive symptoms should not be treated with triiodothyronine T3 and are not reversible. Our objective was to examine if treatment with sustained-release (SR-T3), in patients with normal TSH values and low body temperatures, reduced symptoms consistent with low metabolic rate—and if improvement in symptoms remained even after discontinuation of the T3 treatment regimen.
INTERVENTIONS:
We report the results of 11 euthyroid outpatients with low body temperatures who took oral SR-T3 every 12 hours according to a cyclic dosing schedule designed to titrate low body temperatures to normal. The SR-T3 they took was formulated with the aim of delivering the T3 gradually over 12 hours.
RESULTS:
All of the patients in this study responded with an elevation in oral temperatures and improvement in five symptoms measured following the T3 treatment regimen.
CONCLUSION:
Patients exhibiting hypometabolism with normal TSH levels may experience a reversal of symptoms with normalization of low body temperatures on administration of a sustained-release T3 regimen such that their symptoms remain improved after the regimen has been discontinued.
CLINICAL PEARL PAPER:These are opinion articles written by clinicians who have had successful clinical experience. Although the protocols discussed are not derived from double-blind studies the articles offer clinicians valuable practical clinical skills that can be helpful in treating their patients effectively. |
INTRODUCTION
DW pioneered the use of sustained-release triiodothyronine, especially in the treatment of reversible hypometabolic symptoms. The current study utilizes a protocol (Wilson’s T3 protocol, or WT3 protocol) developed by DW, which involves the patient undertaking one or more cycles of gradually-increasing doses of SR-T3 (q 12 hours orally) according to patient response; in this study the dosing is followed by weaning off the SR-T3 treatment (as is typical). The objective of the WT3 protocol is to normalize low body temperature, thereby reversing hypometabolic symptoms in patients, and ideally for the increase in body temperature and improvement in symptoms to remain even after discontinuation of the SR-T3 regimen.
Hypothyroidism causes symptoms of hypometabolism due to a thyroid hormone deficiency as determined by a low TSH lab test result. However, in DW’s experience, some euthyroid patients have low body temperatures and hypometabolic symptoms that are reversible with SR-T3 treatment. If some euthyroid patients do have reversible low body temperature and hypometabolic symptoms the etiology of said low body temperature and hypometabolic symptoms is not known and may be multi-factorial.
Alleviating hypometabolic symptoms in euthyroid patients may be viewed as analogous to alleviating the symptoms of dysfunctional uterine bleeding. Dysfunctional uterine bleeding is often a diagnosis of exclusion by medical providers based largely on the patient’s report of irregular bleeding, and it may be reversible with a short-term, therapeutic trial of oral contraceptives (and with the patient weaned off the oral contraceptives on normalization of menstrual cycles and attendant elimination of dysfunctional bleeding symptoms). Likewise, with the exclusion of other causes of low body temperature and hypometabolic symptoms patients can be considered for a therapeutic trial of SR-T3. Such symptoms may be reversible upon normalization of body temperatures with a short-term, therapeutic trial of SR-T3 (and with the patient weaned off the SR-T3).
Thyroid system disorders of diverse iterations have been investigated for numerous years. Two examples are “euthyroid hypometabolism” and “hypometabolism without myxedema”. Since before the 1950s, thyroid disorders such as these have been associated with lack of clinical improvement in hypometabolic symptoms, even with the administration (to the point of toxicity) of large doses of thyroid-targeted pharmaceutical agents. Likewise, resistance to thyroid hormone (RTH)—a rare syndrome—is characterized by high T4 and T3 levels with normal (rather than suppressed) TSH levels, as would usually be anticipated. RTH is due to a genetic mutation of the thyroid hormone receptor.
Subclinical hypothyroidism is defined as a high TSH with normal T4 levels. Approximately 2-5% of all patients with subclinical hypothyroidism will progress to overt hypothyroidism per year. Euthyroid Sick Syndrome (ESS) is characterized by an adaptive, temporary change in lab test results (with increased reverse triiodothyronine (RT3), and decreased triiodothyronine (T3) (Fig 1), but usually normal (T4)) brought on by a non-thyroidal illness; lab values return to normal after the illness has resolved. Patients with ESS do not have hypometabolic symptoms, and thyroid-targeted treatment is therefore unnecessary.
Hypometabolism is not an illness but rather a condition, and is not necessarily problematic in a given patient. But, hypothyroidism can cause persistent and problematic hypometabolic symptoms that can require life-long thyroid hormone treatment. DW postulates that there is a reversible hypometabolic syndrome that can also cause persistent and problematic hypometabolic symptoms that can often be corrected with short-term (several months) treatment with T3. DW submits that the reversible hypometabolic syndrome appears to often be brought on by systemic stress due to illness, injury, or emotional trauma: the defining symptom is a reversible slowing of the metabolism; and that reversible hypo-metabolic symptoms often appear to worsen in stages as the level of systemic stress increases. Using the WT3 protocol, the reversible hypometabolic syndrome might be corrected by normalizing the body temperature using T3—even when thyroid values are in the normal range. It is also conceivable that a patient could have such a syndrome and hypothyroidism (or even hyperthyroidism) simultaneously.
WT3 protocol treatment is rejected by the American Thyroid Association (ATA) because no double-blind study has yet been published to support the existence of reversible hypometabolic symptoms. Consequently, the ATA has not approved the use of T3 (and considers the use of T3 to be inappropriate treatment) in the presence of normal thyroid blood tests. However, the diagnosis and treatment of reversible hypometabolic symptoms by normalizing low body temperature has been taught in naturopathic medical schools for years, and studies have been undertaken using T3 in patients with normal TSH levels for over 60 years. DW describes the reversible hypometabolic symptoms as essentially the same symptoms as those of hypothyroidism-induced hypometabolism.
T3 RESEARCH
As with the reversible hypometabolic symptoms, a systemic stressful event is thought to trigger chronic fatigue syndrome (CFS). Meanwhile, fibromyalgia syndrome (FMS) also shares many symptoms in common with CFS. Precedence for T3 treatment of reversible hypometabolic symptoms can be found in recent FMS research; one study found that 75.3% of FMS patients experienced decreased tender-point sensitivity (as measured by algometry) after treatment with 75-150 mcg of T3 in conjunction with other lifestyle changes (i.e., increased aerobic activity, a change in diet, and ingestion of nutrition supplements). Additionally, in Dr. L. Sonkin’s study, 88 euthyroid patients experiencing fatigue, myofascial pain, and depression were placed on thyroid therapy. Dr. Sonkin evaluated the symptoms of each patient before and after the thyroid therapy. He reported that T3 improved the symptoms in many of these patients (Table 1)

EUTHYROID HYPOMETABOLISM SYMPTOMS SCORE
A study of Bunevicius et al. of hypothyroid patients showed that mood, neuropsychological function, and cognitive abilities were much improved in those who had taken T3 as opposed to T4 (levothyroxine). These hypothyroid patients with euthyroid TSH and T4 blood values were already taking exogenous T4, and had hypothyroid cognitive symptoms that resolved with T3 therapy. A conclusion was that the T3 impact on human physiology was greater than generally presumed.
The possibility that a subtle thyroid defect may occur secondary to systemic stress has not been fully explored. However, it is well-documented that— under periods of physical injury as well as chronic or acute illness—a decrease occurs in conversion of T4 to T3. Stress that stimulates the hypothalamic-pituitary-adrenal (HPA) axis results in an increased level of cortisol.In turn, increased cortisol levels are well-known to inhibit the conversion of T4 to T3, resulting in increased RT3. The deiodinase enzymes can locally increase or decrease thyroid hormone signaling in tissue- and time-specific fashion, independent of changes in thyroid hormone serum concentrations and appear to play a much broader role in energy homeostasis than once thought. And, since RT3 has been shown to inhibit type II deiodinase which is the major activating enzyme of T4 to T3 it is conceivable that transient increases in RT3 due to cortisol could secondarily inhibit type II deiodinase, leading to more RT3, thereby contributing to a persistent dysregulation affecting energy homeostasis. This might be the case even though RT3 has a short half-life and is quickly broken down. Whatever the mechanism, it is the clinical experience of DW that the reversible hypometabolic symptoms tend to come on or worsen under conditions of systemic stress often persisting after the stress has passed. Similarly, recent reviews discuss stress as a possible trigger in other conditions such as autoimmune disease and fibromyalgia.
Most research conducted on the use of T3 in patients with normal TSH levels have been performed in patients undergoing treatment for depression. A well-documented relationship exists between disorders of the thyroid axis and depressive symptoms. Because of this relationship to depression, most T3 studies have involved therapeutic trials of thyroid hormones in patients diagnosed with depression along with normal TSH levels. The existence of a well-established causal relationship has also been demonstrated between the thyroid axis function and body temperature. For example, thyroid storm may cause fever, while myxedema may cause low body temperature. Thus, this causal relationship may justify therapeutic trials with T3 to normalize body temperatures in the same manner as therapeutic trials have been justified for T3 treatment of depression. Similar justifications could be made for the use of a therapeutic trial of T3 for other symptoms that might be related to hypo-metabolism in euthyroid patients.
Depressive disorders are diagnosed based on clinical criterion and are often treated with therapeutic trials of antidepressants. Antidepressants have recently become the most commonly prescribed class of medications in the United States. Studies have shown that up to 50% of patients with treatment-resistant depression (on tricyclics or SSRIs) will achieve remission if T3 (25-50 mcg each day) is added to their treatment regimen.
A justification for a trial of innate thyroid hormones may be similar, therefore, to the justification for trials of antidepressants to relieve symptoms. Similarly, bipolar patients have also been shown to respond well when T3 was added to their pharmacologic treatment regimen, even after a remission failure on an average of 14 different antidepressant trials. However, one-third of these bipolar patients achieved complete remission by the addition of T3
It may be beneficial in some cases to prescribe T3 upon clinical criterion as a therapeutic trial just as antidepressants and many other medications are. Using T3 to treat hypometabolic symptoms in euthyroid patients may be appropriate especially considering that numerous studies have also shown that high dose T3 therapy in the treatment of euthyroid depression is often well-tolerated and for long periods of time (e.g., a two-year period).Overall, T3 has been found to be a viable, safe, inexpensive and effective treatment in euthyroid patients with depression. Hypothyroidism can lead to hypometabolic symptoms such as depression. If T3 can often be effective in the treatment of depression, maybe it can sometimes be effective in the treatment of other symptoms that might be consistent with hypometabolism, as well. It is recognized that the HPT axis is responsible for the maintenance of homeostasis of body metabolism. Likewise, hormones and enzymes responsible for hormone synthesis and degradation, along with hormone transporters and receptors, may become dysfunctional necessitating corrective therapeutic intervention. Researchers have documented a sub-group of people diagnosed with depression who may benefit specifically from the use of T3 (as opposed to T4), and suggested T3—as opposed to T4—be administered to euthyroid patients with depression whereas endocrine patients are routinely prescribed T4.
Endocrine disorders have long been recognized to cause clinical psychiatric symptoms; these psychiatric symptoms often resolve when the endocrine disorder is corrected. This has led to increased research investigating a hormonal etiology for a variety of psychiatric disorders. However, much of this research has been unsuccessful in terms of outcome. Meanwhile, research has shown that treatment with T3 in addition to anti-depressants is more successful in some patients whose symptoms were not alleviated by anti-depressants alone. However, it cannot be ruled out that these patients may have responded well to initial T3 monotherapy. No studies have been undertaken yet on the treatment of depression by T3 alone as a medicinal intervention. Since T3 is not patentable, there is little incentive on the part of pharmaceutical companies to fund studies on the use of T3 for the treatment of depression.
One critical factor missing in most published research on T3 as a pharmacological intervention is its effect on body temperature. Research has shown that the administration of T3 increases body temperature in the rat. But in the clinical experience of DW, the dosage and administration of T3 necessary to normalize body temperatures varies exceedingly between individuals but the body temperatures that correlate with improvement in symptoms seem to be remarkably similar from person to person. It may be that body temperature may be a useful guide in the use of T3 to alleviate symptoms. The present study focused on the relationship between T3 dosing according to body temperature (according to the WT3 protocol) and the mitigation of symptoms. Therefore, studies in which research subjects receive the same dose and administration of T3 are not likely to achieve normalized body temperature, and may increase the chance of side effects in those research subjects.
While therapeutic administration of T3 to research subjects has occurred, the T3 administration has not been in accordance with the individual’s body temperature. DW feels that body temperature can be an essential factor in the use of T3 to correct symptoms. Though the use of T3 has been studied in euthyroid patients, our research focused on the relationship of T3 dosage to body temperature for the mitigation of symptoms according to the WT3 protocol.
LOW BODY TEMPERATURE
Body temperature is controlled by a thermoregulatory system that modulates heat production and heat loss, such that core temperature is maintained within a narrow range. The thermoregulatory center of the brain is located in the pre-optic region of the anterior hypothalamus (POAH). Neurons in the POAH act as a thermostat, and modulate heat production and heat loss to control the body temperature. Heat is produced from both metabolic and physical activities.
In a neutral environment, human metabolic rate produces enough heat to maintain an oral temperature of 37°C (98.6°F); thermoregulatory mechanisms become active when heat content increases or decreases.
Due to daily circadian variations, normal body temperature ranges generally fluctuate from a low of 36.1° C (97° F) that usually occurs between 2:00 and 6:00 A.M. to a high of 37.8°C (100°F) that usually occurs between 5:00 and 7:00 P.M. Since many factors affect the thermoregulatory control, patients who have low average body temperatures do not necessarily suffer from hypometabolic symptoms.
MATERIALS AND METHODS
RESULTS
Table 2 presents the results of the study described in the Methods section. (A value of ‘10’ represents greatest severity of symptoms, while ‘1’ represents least severity. A value of ‘0’ represents an absence of symptoms.) Each patient required a different amount of time to achieve normalization of his or her body temperature; recovery time varied between 3 weeks and 12 months.
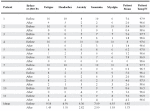
Table 2CFS SYMPTOM SCORE WITH WTS PROTOCOL
Michael Aikin, PhD, conducted a medical statistical analysis of these results (Table 3). His statistical analysis showed statistically significant reductions in the mean symptom scores representing an improvement in the symptoms. After the WT3 treatment was discontinued (usually 3 to 6 months after initiation), the majority of reversible hypometabolic symptom patients reported significant and continued improvement in their symptoms. Notice that 3 of the patients reported almost complete resolution of fairly severe symptoms (Table 2) Additionally, 8 out of the 11 patients reported significant and continued improvement in their symptoms at 30 day follow-up.

Table 3 STATISTICAL ANALYSIS OF SYMPTOMS SCORES WTS Protocol In CFS Patients
DISCUSSION
WT3 PROTOCOL:
The aim of the WT3 protocol is to normalize low body temperature in a manner such that normalized body temperature can be maintained even after treatment by T3 has been discontinued. This is analogous to use of oral contraceptives in the treatment of dysfunctional uterine bleeding to re-establish normal menstrual cycles—such that normal cycles will persist following oral contraceptive discontinuation. One of the co-authors of this article DW developed the WT3 treatment protocol over a two-year period based on his empirical observations in treating patients with intractable fatigue. Wilson was the first physician to utilize sustained-release T3 therapy in this area; he treated approximately 5,000 patients successfully over a period of four years. A significant number of medical physicians worldwide currently use this protocol in their clinical practices as a therapeutic method.
UNIQUE FEATURES OF WT3:
Four unique features of the WT3 protocol that distinguish it from conventional T3 therapy are that: a) it is often used only temporarily as a means to recalibrate body temperature patterns; b) it is often used even when TSH tests are completely normal; c) it uses a sustained-release form of T3, as opposed to non-sustained release liothyronine (Cytomel®, Jones Pharma) d) it is cyclic in its administration, with the dose and cycle-length adjusted according to patient response in symptoms and temperature.
In DW’s clinical experience, a dose escalation approach aimed at normalizing a patient’s temperature with T3 is often helpful in reversing symptoms. In utilizing his protocol, DW usually starts a patient on a low initial dose (i.e. 7.5 mcg of ST-T3 every 12 hours), but may increase the dose by 7.5 mcgper dose per day if average oral temperature remains less than 98.6° F— and the T3 dose can even be increased up to 90 mcg every 12 hours, if necessary. If the body temperature still remains low, the patient may need to be weaned off the T3 by 7.5 mcg decrements per dose approximately every 2 days (or slower, to keep the temperature from dropping) until discontinuation, and then commence another cycle entirely. The significance of cycling on and off escalating doses of T3 will be discussed in the following paragraphs.
Overall, long-term success in resetting metabolism and alleviating symptoms using the cyclic SR-T3 therapy proposed for this study appears to be related to holding body temperatures close to 98.6°F on the same dose for a time (up to 3 weeks) suggesting that negative feedback inhibition of the thyroid system has been overcome (DW refers to this as “capturing” the temperature). Patients whose body temperatures have been successfully raised to approximately 98.6°F tend to show a sustained positive response with alleviated symptoms well after the treatment has been discontinued. On the other hand, patients whose temperatures remain low appear to obtain less benefit from the WT3 treatment.
It is known that exogenous T3 suppresses T4 levels due to negative feedback inhibition of TSH, and it is speculated that T4 suppression is an important aspect of the WT3 protocol, essential to its effectiveness in raising body temperature. More than one cycle of SR-T3 therapy may be needed to increase body temperature to within normal range. In many cases, one or more cycles reaching a maximumdose of SR-T3 (90 mcg twice per day) fails to raise the patient’s body temperature to approximately 98.6°F. However, the body temperature may then normalize at a lower dose on a subsequent cycle. In such cases, we speculate that greater T4 suppression has been achieved with each successive cycle until the body temperature rises to within a normal range. The number of cycles implemented varies between patients but usually ranges between one to five. Serial measurements of serum free T4 may help to elucidate this proposed mechanism.
We also speculate that T4 suppression from repeated cycles of SR-T3 therapy results in lower serum levels of T4 and RT3, and therefore less competitive inhibition of T3. Also, with lowered RT3 levels, there may be less competitive inhibition against endogenous T4 by RT3 at the 5’-deiodinase enzyme that converts T4 to T3. This may partly explain why progress in symptom alleviation, body temperature increase, and increased T4 suppression may occur (perhaps at lower T3 dosage levels) from cycle to cycle (with increased T3 expression during each cycle). Increased expression of T3 may result in even further T4 suppression, until the body temperature is finally normalized and the metabolism is “re-calibrated.” Once the temperature is normalized (captured), decreased dosages of SR-T3 tend to be needed on subsequent cycles to maintain normal body temperature. Regardless of the mechanism, in our experience WT3 therapy seems to aid metabolism in the vast majority of euthyroid patients with hypometabolic symptoms. Our experience and data suggest that patients can gradually discontinue the SR-T3 therapy and often retain its benefits, once their body temperatures stabilize at approximately 98.6° F through one or more cycles of T3 therapy per the WT3 protocol.
The 11 subjects in this study were treated with liothyronine compounded in a hydrophilic matrix system, employing hydroxypropyl-methylcellulose (HPMC) designed to be taken every 12 hours. The liothyronine is synthetically made and does not differ from any standard pharmaceutical preparation of T3, except that it is compounded with the sustained release agent, methylcellulose. Interestingly, clinical observation using SR-T3 shows that by cycling on and off T3 the patient’s susceptibility to the pharmaceutical effects of the T3 can change from cycle to cycle. (For example, the first cycle of SR-T3 may require a dose of 90 mcg of T3 twice per day to obtain a normal temperature, while the second cycle may require only 45mcg of T3 twice per day to achieve the same body temperature.) Initially, the protocol used liothyronine sodium (Cytomel® Jones Pharma), but undesirable side effects (i.e.,irregular heartbeat and occasional atrial fibrillation) prompted a change to the use of SR-T3 exclusively. Arial fibrillation has been rare with the use of SR-T3. However, a significant number of patients still suffer from side effects related to vacillating T3 levels. These side effects have included heart palpitations, increased heart rate, irritability, shakiness, fatigue, and headaches. However, these side effects are usually successfully treated with a “test dose” of T4 of .0125-.025 mg of levothyroxine to dampen the effects of T3 therapy through inhibition of T3 by T4. T4 has 25% of the potency of T3; In DW’s experience administration of T4 usually decreases or eliminates the side effects of T3 therapy within 45 minutes.
DW’s experience suggests that T3 compounded in a hydrophilic matrix system (sustained-release system), taken every 12 hours in capsules, provides a predictable, well-tolerated method to normalize body temperature patterns and alleviate the hypometabolic symptoms in some euthyroid patients.
An additional important consideration related to proper thyroid hormone function in patients is iodine consumption, absorption and utilization. Iodine deficiency can result in thyroid function abnormalities and, when severe, can result in endemic goiter and cretinism, endemic mental retardation, decreased fertility rate, increased perinatal death, and infant mortality. Iodine can also be important in fibrocystic breast disease and breast cancer. Iodine deficiency should be evaluated to avoid secondary physiological deregulation and its associated risk factors. If deficient, iodine supplementation is indicated.
Currently, there is some controversy over the effects of thyroid hormone supplementation on osteopenia. Most studies indicated no effect.However, one study of long-term suppressive doses of T4 did show an increase in bone loss. Clinical studies using supraphysiological doses of T3 (ranging from 93.75 to 105 mcg daily) in euthyroid fibromyalgia over an 8-month period indicated no change in serum calcium and phosphorous—nor in bone densitometry versus placebo. On the other hand, there were higher levels of urinary N telopeptides. which might suggest increased bone resorption. Liver function tests revealed no difference versus placebo at 4-month follow-up; serum creatinine and calcium were also normal, indicating no change in muscle mass. Mean heart rate significantly increased from 68.5 bpm in placebo, and in T3 patients from 83.94 bpm. No patient developed tachycardia, and there was no significant difference in diastolic or systolic blood pressure in T3 or placebo group.
Adverse reactions to liothyronine sodium can be due to therapeutic over-dosage, and thus can mimic the typical symptoms of hyperthyroidism (e.g.,fever, tachycardia, irritability, nervousness, and increased bowel motility). However, similar side effects along with normal or low body temperatures can be due to unsteady T3 levels that can arise from the dosing or delivery of T3 which has a short half-life. In rare instances, allergic skin reactions have been observed in patients taking liothyronine sodium.
Based on our promising clinical results, the author of this article believes euthyroid patients may benefit from increased utilization of the WT3 protocol as a treatment modality.
DISCLOSURE OF INTERESTS
E. Denis Wilson is president of the Muskeegee Medical Publishing Co., Lady Lake, FL, co-founder of WTSmed, Inc. Montpelier, VT, and a Consultant for Medaus Pharmacy, outside the submitted work.
REFERENCES
- Merck Manuals.
- Davis A, Godwin A, Lippman J, et al. Triphasic Norgetimate-ethinyl estradiol for treating dysfunctional uterine bleeding.
- Fraser IS. Treatment of ovulatory and anovulatory dysfunctional uterine bleeding with oral progestogens.
- Kurland GS, Hamolsky MW and Feedberg AS. Studiesin nonmyxedematous hypometabolism: The clinical syndrome and the effects of triiodothyronine, alone or combined with thyroxine.
- Refetoff S, Weiss RE and Usala SJ. The syndromes of resistance to thyroid hormone.
- McDermott MT, et al. Subclinical Hypothyroidism Is Mild Thyroid Failure and Should be Treated
- Gillet M. Subclinical Hypothyroidism; Subclinical Thyroid Disease: Scientific Review and Guidelines for Diagnosis and Management.
- Felicetta J. Effects of Illness on Thyroid Function Tests.
- American Thyroid Association. http://www.thyroid.org/professionals/publications/statements/99_11_16_wilsons.html 2005.
- Fields EM. Metabolic Stimulants-Reply,
- Morton JH. Sodium Liothyronine in Metabolic Insufficiency Syndrome and Associated Disorder Prelim report.
- Tittle RC. Effects of 3,5,3; L-Tri Iodothyronine in Patients with Metabolic Insufficiency Prelim Report.
- Lam K.S., et al., Vasoactive intestinal peptide in the anterior pituitary is increased in hypothyroidism.
- Karkal S. Overcoming diagnostic and therapeutic obstacles in hypothyroidism.
- Lowe JC, Garrison RL, Reichman AJ, Yellin J, et al. Effectiveness and safety of t3 therapy for euthyroid fibromyalgia: a double-blind placebo-controlled crossover study.
- Buchwald D, Garrity D. Comparison of patients with chronic fatigue syndrome, fibromyalgia, and multiple chemical sensitivities.
- Lowe JC, Reichman AJ, Yellin J. The process of change during T3 treatment for euthyroid fibromyalgia: A double-blind placebo-controlled crossover study.
- Sonkin LS. Myofascial pain due to metabolic disorders: diagnosis and treatment in Myofascial pain and Fibromyalgia: Trigger Point Management. Rachlin, ES (Ed.), 1994, Mosby: St Louis, MO, pp. 45–60.
- Bunevicius R, Kazanavicius G, Zalinkevicius , et al. Effects of thyroxine as compared with thyroxine plus triiodothyronine in patients with hypothyroidism.
- Schimmel M and Utiger RD. Thyroidal and peripheral production of thyroid hormones. Review of recent findings and their clinical implications.
- Refetoff S. Resistance to thyroid hormone: an historical overview.
- Scott LV, Svec F and Dinan T. A preliminary study of dehydroepiandrosterone response to low-dose ACTH in chronic fatigue syndrome and in healthy subjects.
- Gereben B. Cellular and Molecular Basis of Deiodinase-Regulated Thyroid Hormone Signaling.
- Kaiser CA. In Vivo Inhibition of the 5’-Deiodinase Type II in Brain Cortex and Pituitary by Reverse Triiodothyronine.
- Stojanovich L, Marisavljevich, D. et al. Stress as a trigger of autoimmune disease.
- Martinez-Lavin M. Stress, the stress response, and fibromyalgia.
- Joffe RT. Hormone treatment of depression.
- Cooper R, Lerer B. The use of thyroid hormones in the treatment of depression.
- Olfson M, Marcus SC. National Patterns in Antidepressant Medication Treatment.
- Manning JS. What alternatives to first-line therapy for depression are effective?
- Abraham G, Milev R, Stuart Lawson J. T3 augmentation of SSRI resistant depression.
- Kelly T, Lieberman DZ. The use of triiodothyronine as an augmentation agent in treatment-resistant bipolar II and bipolar disorder NOS.
- Kelly T, Lieberman DZ. Long term augmentation with T3 in refractory major depression.
- Gold MS, Pottash ALC, Extein I. Hypothyroidism and Depression Evidence from Complete Thyroid Function Evaluation.
- Johansson C, Thoren P. The effects of triiodothyronine(T3) on heart rate, temperature and ECG measured with telemetry in freely moving mice.
- Llewellyn-Smith IJ, Verberne AJM.
- Friedman M. Supraphysiological Cyclic Dosing of Sustained Release T3 in Order to Reset Low Basal Body Temperature.
- Wilson ED.
- Ju RT, Nixon PR, Patel MV. Drug release from hydrophilic matrices. 1. New scaling laws for predicting polymer and drugrelease based on the polymer disentanglement concentration and the diffusion layer.
- DeLange F. The disorders induced by Iodine Deficiency.
- Ghent W, et al. Replacement in Fibrocystic Disease of the Breast.
- Smyth PPA. Thyroid Disease and Breast Cancer.
- Van Den Eeden SK, Barzilay JI, Ettinger B and Minkoff J. Thyroid hormone use and the risk of hip fracture in women > or = 65 years: a case-control study.