Abstract
This paper reviews the use of whole-plant Artemisia annua (sweet Annie) as it pertains to malaria including history, chemistry, clinical efficacy, pharmacokinetics, dosing, safety, and resistance. Artemisinin, the sesquiterpene lactone found in the plant, and various semi-synthetic variants of it used as critical drugs around the world for malaria are discussed in comparison to the whole plant. The multifaceted potential of A. annua against malaria including as a mosquito larvicide and to prevent transmission is discussed. The critical importance of combination therapy with this herb (be it with well-studied synthetic drugs or as-yet poorly studied natural products with activity against malaria, notably curcumin) is reviewed. The relevance of non-artemisinin chemicals in A. annua is highlighted. The pharmacokinetic and pharmacodynamic limitations of A. annua and artemisinins for prophylaxis against malaria are presented. Other Artemisia species besides A. annua, whether they contain artemisinin or not, are also touched upon in the context of malaria.
In the past 20 years, an entirely new class of antimalarial agents has become available. They are derived from the sesquiterpene lactone artemisinin, which is found in the medicinal plant Artemisia annua, and its principal metabolite dihydroartemisinin. This article will review the proper of use A. annua, artemisinin and its derivatives (natural and semi-synthetic; see Figures 1 and 2) for treatment of malaria as well as their limitations for primary prevention of malaria.
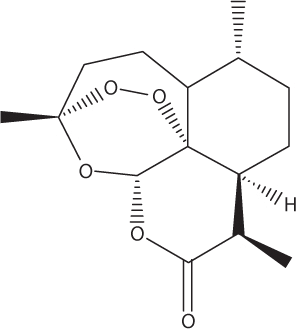
Artemisinin.
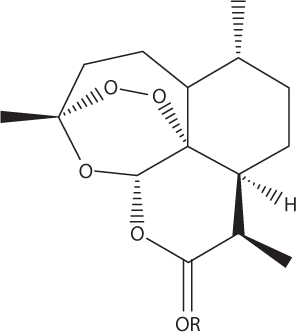
Semisynthetic variants of artemisinin.
Artemether: R=CH3
Arteether: R=C2H5
Artesunate: R=COCH2CH2CO2Na
A. annua is known in English as sweet Annie and in Mandarin as qìng hào (“green hao”). However, one review of the ancient Chinese literature concludes that this name applies better to the closely related artemisinin-containing plant A. apiacea, and that A. annua is more correctly referred to ashuàng huà hào (“yellow blossom hao”). A. apiacea also contains artemisinin but at lower levels than A. annua. The earliest written record of the use ofA. annua comes from the anonymous text known as the Wuˇshíèr Bìngfàng (Recipes for 52 Ailments) discovered in the Mawangdui tomb believed to date from 215 BCE. The first extant, clear, written account of the use of A. annua for malaria occurred in Zhouhou Beiji Fang (The Handbook of Prescriptions for Emergencies) by Ge Hong written in the 4th century CE. The Shennong Ben Cao Jing (circa 400–300 BCE) also mentions A. annua.
A. annua is a fast-growing annual weed that can attain 2 m in height. It can grow in a wide range of soil conditions, though well-drained soil is generally best. The plant is wind pollinated and produces enormous quantities of seeds. The part used therapeutically is the flowering top. A. annua has many chemotypes and cultivars with varying chemistries that can significantly affect medicinal activity. The high-artemisinin cultivars that dominate in China and Africa develop peak artemisinin levels before flowering, but research on low-artemisinin cultivars suggest that their synergistic polymethoxylated flavonoids peak during flowering. A. lancea and A. apiacea have also been reported to contain artemisinin, albeit in much smaller amounts.
In 1972, Chinese scientists isolated qinghaosu (artemisinin) from A. annua. They were looking at this plant very specifically because they took Ge Hong’s writings about qinghao from over 1000 years before seriously (as opposed to many researchers in the west who look upon historical herbal books as unhelpful superstition). Based on in vitro results showing artemisinin was a potent antimalarial, development of this compound as a medicine began in earnest. Initial results with a tablet form of artemisinin were not that promising, apparently due to poor bioavailability. Further research with different doses and formulations led to improved efficacy and the development of several synthetic analogs. These compounds are now recognized the world over as a novel class of effective antimalarials, and they have come into widespread use (see Table 1).

Artemisinins.
MECHANISM OF ACTION
Artemisinin is a sesquiterpene lactone found in A. annua that appears to work in part due to the presence of an endoperoxide bridge. The heme inside schizonts (see Figure 3) breaks the bridge, creating a powerful free radical form of the artemisinin, which then attacks parasite proteins without harming the host. It appears to be a very targeted oxidative agent, and one that is not cyclically oxidized and reduced.
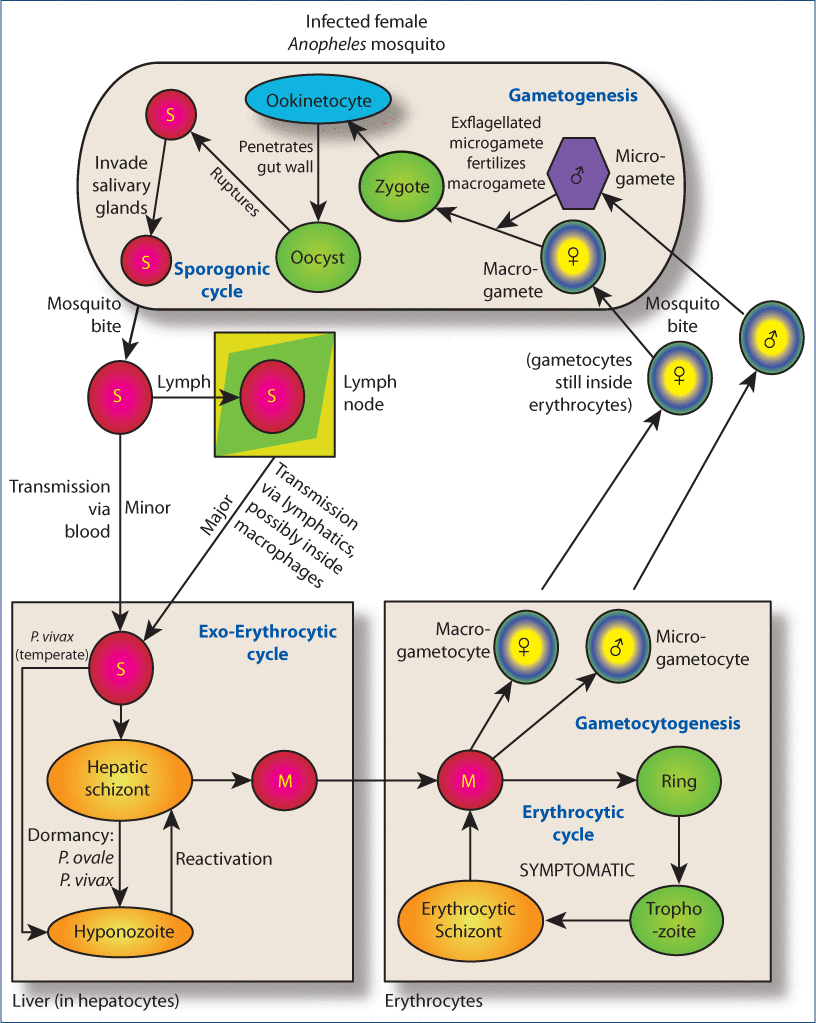
Figure 3:Overview of the life cycle of Plasmodium spp.Legend: When an infected female Anopheles mosquito takes her blood meal, she infects the human host with sporozoites (<200 cells/drop of mosquito saliva are sufficient for infection). These travel, largely through the lymphatic system and minimally through the blood, to hepatocytes. There, most Plasmodium spp rapidly form hepatic schizonts, but the species shown can also form dormant cells called hypnozoites (which can periodically reactivate, cause a relapsing infection). Hepatic schizonts fill with merozoites, then rupture, releasing them into the blood where they infect erythrocytes. Release of a new wave of merozoites from the hepatic schizonts after successful reduction of erythrocytic stages of the organism below detectable levels is known as recrudescent infection. Two things can happen after invasion of erythrocytes. Usually the merozoites establish the erythrocytic cycle, forming immature (ring) trophozoites that develop into mature trophozoites which form erythrocytic schizonts. These fill with merozoites, rupturing, and ultimately the host erythrocyte ruptures releasing the merozoites to infect other erythrocytes. Some merozoites, however, develop into male and female gametocytes. These do not cause pathology in the host but are uptaken by uninfected mosquitoes that take blood meals and thus spread infection. Gametocytes rupture out of erythrocytes when they enter the mosquito midgut and undergo transformation into gametes. After fertilization, a zygote forms. This develops into a motile ookinetocyte which pierces the mosquito’s gut wall and forms an oocyst between the epithelium and basal lamina. This eventually ruptures releasing motile sporozoites that traverse the mosquito and infest her salivary glands, ready to infect a new host at the next blood meal. Artemisinin attacks multiple stages of the erythrocytic cycle as well gametocytes. M, merozoite; S, sporozoite.
Artemisinins actually appear to work against multiple forms of the parasite, not just schizonts, during its erythrocytic cycle – notably rings and trophozoites. As artemisinins target the erythrocytic cycle, they should be most effective at treating acute malaria, which is how they are generally used and where the best support of efficacy exists.
Artemisinin also effectively kills early-stage gametocytes (the form of malaria transmitted from humans to mosquitoes). Therefore, artemisinins may help prevent spread of the parasites from infected humans to uninfected humans. Open trials of artesunate, a semi-synthetic artemisinin, with sulfadoxine, pyrimethamine and the mature gametocyte-killing primaquine in India show that this is fairly effective at reducing gametocytes of P. falciparum. For a review of the four major species of Plasmodium that infect humans, see Table 2. A trial is underway in Uganda using artemether, another semi-synthetic artemisinin, with lumefantrine and primaquine, for reducing or eliminating gametocyte burden. Adding artemisinins to mefloquine led to significant reductions in gametocytemia compared with mefloquine alone. For a review of non-artemisinin antimalarial drugs, seeTable 3.

Major Plasmodium species infecting humans.

Review of available antimalarial drugs.
Artemisinins do not affect persistent hepatic forms of malaria (hypnozoites) seen with P. vivax and P. ovale, therefore artemisinins do not effect a complete cure (eliminate malaria permanently from the body). Only primaquine has been shown to be effective for this purpose. As artemisinins do not affect sporozoites (the forms of malaria injected by mosquitoes into human hosts), they are not effective for preventing infection when taken prophylactically.
Unfortunately some people who are ignorant about the details of malaria or how artemisinins work continue to recommend artemisinin or A. annua for prevention of malaria, often citing anecdotal evidence (if any) that they or people they have recommended it to took it and did not develop malaria. Of course, this could be due to chance and precautions taken against mosquito bites. Given the high likelihood that artemisinin and A. annua have no preventive effect, they are not recommended to be used in place of existing malaria prophylaxis.
Several flavonoids in A. annua, notably methyoxylated flavones such as eupatorin, chrysoplenol-D, chyrsoplenetin, and cirsilineol (see Figure 4), all dramatically enhanced the activity of artemisinin against chloroquine-resistant P.falciparumin vitro. Previous research found a similar result for the flavonoid casticin from A. annua. Casticin, however, is poorly extracted in and rapidly lost from aqueous extracts of A. annua. None of these flavonoids had activity against malaria by themselves. Chrysophenol-D and chrysoplenetin also potentiated the activity of berberine against multidrug-resistant Staphylococcus aureusin vitro. At the very least, this supports the argument in favor of studying a combination of the whole plant or non-artemisinin components of it with pharmaceutical antimalarials to see if they amplify efficacy.
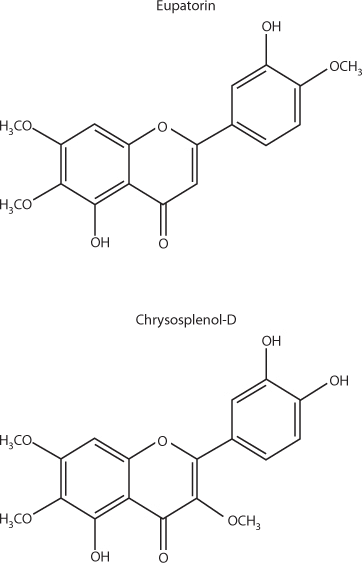
Selected methoxylated flavonoids of Artemisia annua.
Lest it seem that A. annua or artemisinins are active only against Plasmodium, Table 4 summarizes research showing that in fact they are broad-acting antimicrobial agents. Further work should go into determining whether they are actually clinically effective for infections with these agents.

Other reported organisms killed or inhibited by artemisinins.
A. annua and artemisinins likely also have effects on the immune system and inflammation that are potentially relevant in malaria patients and in other conditions. Aqueous infusions of A. annua significantly decreased levels of inflammatory cytokines including interleukin (IL)-6 and IL-8 in human intestinal cells in vitro. Rosmarinic and chlorogenic acids in the herb seemed primarily responsible for this effect. Without causing global immunosuppression, dihydroartemisinin inhibited the mammalian target of rapacmycin (mTOR) pathway, which is associated with higher Treg helper lymphocyte activity and reduced T helper lymphocyte activity in mice with a form of multiple sclerosis. The closely related herb A. apiacea inhibited nuclear factor κB activation in rats, thus limiting inflammation. In vitro, A. capillaris inhibited secretion of the pro-inflammatory cytokines tumor necrosis factor-α and IL-1.
CLINICAL EFFICACY:
Isolated artemisinin and its principal active metabolite, dihydroartemisinin, have been shown to be quite effective at curing acute uncomplicated and falciparum malaria according to meta-analyses of clinical trials. The studies below look at whether whole-plant A. annua products are also effective.
Two clinical trials have assessed the efficacy of oral administration of an infusion of a high-artemisinin (0.5–0.75%) cultivar developed in the Netherlands known as A. annua cv Artemis. In the initial, uncontrolled pilot study, 44 (92%) of 48 African patients (in the Democratic Republic of the Congo) with chronic malaria had total parasite clearance, and 37 (77%) were asymptomatic after 5 days of treatment. A total of 12 (25%) of the patients suffered nausea during treatment, not surprising with such a bitter plant. Nausea cleared with termination of therapy. The total dose of artemisinin delivered on average was approximately 60 mg, far below the usual dose recommendations of purified artemisinin (500–1000 mg a day for 3–5 days). It is possible other compounds, such as the flavonoids mentioned above, in the whole herb potentiated the action of the small amounts of artemisinin, or that small doses of this compound are more effective than previously believed. Note that in vitro research supports this idea, with A. annua infusions significantly inhibiting P. falciparum at concentrations of artemisinin far below those considered clinically effective.
In a follow-up trial, 132 non-pregnant adult patients with P. falciparum infection in the Democratic Republic of the Congo were randomly assigned to receive A. annua lower-dose tea (5 g herb/1 L water/day in four divided doses), A. annua higher-dose tea (9 g herb/1 L water/day in four divided doses), or quinine sulfate 300 mg three times daily for 7 days. The same Artemis cultivar mentioned in the previous trial, cultivated in the Democratic Republic of the Congo, was used in this trial. Artemisinin content was determined to be 1.4% in the herb used, yielding daily doses of artemisinin in the tea groups of 47 mg and 94 mg respectively. In the lower-dose tea group, the 7-day cure rate (defined by negative blood films) was 77% (30/39) compared with 70% (23/33) in the higher-dose tea group and 91% (39/43) in the quinine group. Day 35 cure rates were 34% (11/32) in the low-dose group, 30% (9/30) in the high-dose group, and 79% (27/34) in the quinine groups, indicating very high recrudescence rates in the artemisinin groups. Adverse effects were comparable among groups and not different from symptoms of malaria, except for a 27% tinnitus incidence in the quinine group. This trial suggests that A. annua tea by itself is fairly effective for immediate relief of falciparum malaria but is not effective at keeping the infection under control and needs to be combined with other treatment to prevent recrudescence and, possibly, development of resistance. This concurs with standard use of artemisinins which are always recommended for use in combination therapy and not as monotherapy. Research has not been published on the question of whether resistance can occur to whole-plant A. annua.
In a small randomized, double-blind trial conducted in 19 falciparum malaria-infected Tanzanian adults, an infusion of A. annua (cultivar and dose unknown) was compared to sulfadoxine-pyrimethamine monotherapy. The 7-day cure rate was 70% (7/9) for the tea and 78% (7/9) for the drug. The 28-day cure rate was 11% (1/9) for the herb and 38% (3/8) for the drug. Thus these two treatments appear to have similar immediate beneficial effects but unacceptable high recrudescence rates.
These proof-of-concept trials suggest that a sustainable supply of locally grown sweet Annie could be maintained in Africa as a way to deliver life-saving medicine, and that pharmaceutical companies and synthetic, isolated artemisinins are not necessary for treatment of malaria. However, they also show that crude extracts of sweet Annie alone are unlikely to be effective at preventing recrudescence and so some pharmaceutical preparations will need to be combined with them. A critical question is whether combination herbal medicine could allow for a truly sustainable approach to malaria treatment. It is not yet known what the best drugs to combine with A. annua would be, but logically it would be those with radically different molecular targets than artemisinin, such as anti-folate drugs (e.g., sulfadoxine-pyrimethamine) or 4-aminoquinolines that target heme degradation (e.g., chloroquine). In areas with high P. vivax or P. ovale infection rates, combination with the anti-hypnozoite drug primaquine is also potentially beneficial.
A. annua also has the potential to be a mosquito larvicidal and adulticidal agent. A hexane extract was moderately effective with a 244.5 ppm LC50against the mosquito vector Anopheles sinensis, 276 ppm against the dengue vector Aedes aegypti, and 375 ppm against the arbovirus and avian malaria vector Culex quinquefasciatus. A petroleum ether extract of A. annua had an LC50 of 78.2 ppm against C. quinquefasciatus mosquito development and growth in prior study.A. annua chloroform extracts had an LC50 of 18.5 ppm against A. sinensis larvae in another study. Larvae of A. stephensi, another malaria vector, were killed at an LC50 of 20 ppm. Thus A. annua could potentially be grown locally not only as an acute schizonticidal agent for rapid control of malaria symptoms but also as a way to control malaria mosquito vectors.
Species of Artemisia not containing artemisinin, or at most tiny concentrations, are antimalarial in preclinical studies (see Table 5). This is presumably due to the presence of other antimalarial compounds, which may or may not be similar to artemisinin in structure. It is also likely that they contain non-antimicrobial compounds like the methoxylated flavonoids of A. annua that enhance efficacy by reducing resistance or via other mechanisms. This hypothesis needs verification urgently, as it could provide new medicines for malaria.

Other Artemisia species reported to have anti-plasmodial activity (regardless of artemisinin content).
COMBINATION HERBAL THERAPY:
The most critical antimalarial medications have been derived from natural products, primarily quinine and artemisinin. There is a growing awareness that herbs have a role to play in the global fight against malaria. The approach advocated by this author is to use a range of herbs to optimally attack malaria parasites with multiple chemicals against multiple pathways, and thereby minimize the already low (and purely theoretical) risk of resistance but also toxicity (by limiting the absolute dose of each herb necessary for efficacy).
Suggestions for sustainable combination therapy with A. annua using local herbal resources, or plants that are likely sustainable in many tropical climates, include Cinchona spp (Peruvian bark and the source of quinine and related antimalarial alkaloids), Azadirachta indica (neem) leaf or oil,Vernonia amygdalina (bitter leaf), Argemone mexicana (Mexican poppy), and Curcuma longa (turmeric). Other herbs proven effective in clinical trials may also be relevant in the specific geographic regions in which they grow (see Table 6).

Antimalarial whole plants in preliminary clinical trials.
Clinical trials have previously demonstrated that a multiple alkaloid extract of Peruvian bark is effective for patients with chloroquine-resistant malaria. The rationale for returning to a whole-plant approach to Peruvian bark has previously been outlined, similar to the arguments presented here for sweet Annie. Lower doses of quinine are generally delivered by whole-plant extracts than are used pharmacologically, but the synergy of multiple constituents apparently still allows for clinical efficacy.
Neem has not yet been shown to be effective for treating malaria patients in clinical trials. In animal trials, it is clearly antimalarial, including killing gametocytes which should help reduce malaria spread from infected people to new hosts. Empirically, an infusion of neem leaves has been effective at preventing and treating acute malaria, at least in some patients in Kenya. However this is not proof of efficacy and clinical trials are needed. Neem extracts are also antiretroviral, which could prove important given the continuing rise of human immunodeficiency virus (HIV) and malaria co-infection in many parts of the developing world. Finally, neem oil is an effective mosquito repellant against Anopheles mosquitoes and anAnopheles mosquito larvicide, making this a multifunctional plant.
Curcuminoids, a mixture of yellow, liphophilic molecules found in Curcuma longa (turmeric) often referred to as just curcumin, show promise at enhancing the antimalarial potency of artemisinins. In P. berghei-infected mice, a combination of 100 mg/kg of body weight of curcumin orally after a single intramuscular injection of arteether led to 100% survival of the mice, compared with 100% mortality with arteether injection alone after 11–34 days. Liposomal microencapsulation of artemisinin was just as effective as artemisinin combined with curcurmin in mice infected with malaria, although the combination of the two was dramatically more effective than non-liposomal artemisinin alone at preventing mortality. Another study found that curcumin liposomal formulations combined with arteether prevented mortality and recrudescence in malarious mice, and were significantly more effective than curcumin by itself. Other studies combining curcumin and arteether suggest that through immune mechanisms, curcumin prevents recrudescence. The use of curcumin does appear to have limitations. Curcumin did not increase the efficacy of artemisinin against artemisinin-resistant P. chabaudi in mice. The addition of piperine, an alkaloid from Piper longum (long pepper) and P. nigrum (black pepper), which increases absorption of curcumin, also did not help in this resistant clone.
Some other herbs that have been reported in preliminary human trials to effectively combat malaria, and thus could be considered in combination herbal therapy, are listed in Table 6. There is an urgent need to determine whether combinations of these herbs with A. annua and other antimalarial herbs is effective acutely and at preventing recrudescence, as well as at preventing spread of malaria.
RESISTANCE:
As has been the case with all single chemical entity antimalarial drugs, use of semisynthetic artemisinins has led to the rise of malarial resistance to these drugs. In Cambodia, resistance to dihydroartemisinin already appears widespread. It is unclear if resistance develops to whole-plant extracts or not. This is unlikely as they contain multiple compounds and not a single agent, including the aforementioned methoxylated flavonoids that appear to prevent development of resistance. Ongoing searching of the literature by the author has still failed to find a single paper documenting development of resistance of microbes to whole plants or complex plant extracts. In fact, most papers that have looked at whole-plant extracts have found that they do not induce resistance; for example, Hypericum perforatum (St. John’s wort) does not induce resistance in Staphylococcus aureus, while its isolated constituent hyperforin can. Melaleuca alterntifolia (tea tree) volatile oil, which is a semi-refined extract in that it contains no water soluble compounds, has been shown to be able to induce resistance in Staphylococcus aureusin vitro.
DOSING:
A. annua and artemisinins are not recommended for use as monotherapy in order to prevent resistance developing, and because recrudescence is so high following monotherapy. Therefore, the doses presented here apply only when the agents in question are combined with other treatments.
|
Adult doses for crude A. annua Dried crude herb, 2–4 g tid–qid Dried herb infusion, 1.5–3 g/250 mL water, steeped 15 min, 250 mL 4x/d Fresh herb tincture, 60–90% ethanol, 1:3 weight:volume, 5–10 mL 4–6 x/d These doses should be continued for 5 days (in women) or 7 days (in men) then discontinued (as absorption drops off dramatically after that point). Notes: Dried herb should not be decocted for more than a few minutes, this is associated with rapid destruction of all artemisinin. Doses for children should be proportionally reduced based on body size. A. annua should not be used as monotherapy. |
Presently, the World Health Organization (WHO) recommends using only the semi-synthetic derivatives of artemisinin known as dihydroartemisinin (though humans convert artemisinins to this metabolite), artemether and artesunate for uncomplicated malaria. Though these drug combinations have been documented to cure acute malaria patients effectively, it is questionable whether continuing a synthetic pharmaceutical approach will be sustainable ecologically or economically. The historical record suggests it probably won’t be, if one looks at the high rate of resistance to semi-synthetic quinine derivatives around the world. Quinine is an alkaloid from various species of the Amazonian tree Cinchona spp.
Mixing dried A. annua leaf into millet porridge and cooking them together has been shown to be a palatable and stable delivery form for the herb to children in Kenya. Pharmacokinetic and clinical efficacy profiles of this delivery form have not been published. Further research is warranted given the sustainability of this approach.
|
WHO Guidelines for synthetic artemisinins combination therapy Dihydroartemisinin 4 mg/kg bw for 3 d + piperaquine 18 mg/kg bw for 3 d, but no registered/quality form available Artemether (20 mg) + lumefantrine (120 mg), 1–4 caps bid for 3 d (with fat; worldwide) Artesunate 4 mg/kg bw + amodiaquine 10 mg base/kg bw qd for 3 d (worldwide) Artesunate 4 mg/kg bw qd for 3 d + mefloquine 25 mg base/kg bw qd for 2–3 d (Asia only) Artesunate 4 mg/kg bw qd for 3 d + sulfadoxine-pyrimethamine 25/1.25 mg base/kg bw single dose |
PHARMACOKINETICS:
Artemisinins have short half-lives, certainly <60 min. Dihydroartemisinin, which all artemisinins covert to, has a half-life of around 60 min. This is a significant part of why recrudescence is so high with artemisinins; the compounds rapidly kill the parasites down to an undetectable level (providing immediate and important clinical relief), and then the artemisinins are cleared quickly so the chemicals don’t have time to completely eliminate them. Within a few days of finishing treatment, the small numbers of residual parasites can reproduce and create recrudescent disease. Use of longer-acting drugs combined with artemisinins is essential to long-term efficacy in patients with malaria.
The pharmacokinetics of artemisinin from a tea made from A. annua cultivar Artemis has been studied in humans. The maximum amount of artemisinin was extracted by covering 9 g of dried leaf with 1 L of boiling water and allowing it to sit, away from heat, covered, for 10 min before filtering and pressing. This yielded 94.5 mg of artemisinin with 76% extraction efficiency. Fourteen European adults then drank 1 L of this tea over an average of 15 min, then had their serum artemisinin concentrations determined serially over 8 h. Maximal serum concentration (Cmax) averaged 240 ng/mL and the time to achieve this (tmax) was 0.6 h on average. Area under the curve (AUC) averaged 336 ng/mL×h. The elimination half-life (t1/2) averaged 0.87 h. For comparison to pharmacokinetic studies of pure artemisinin in capsules, see Table 7.

Pharmacokinetics of artemisinin from Artemisia annua compared to pure artemisinin.
Note that the tea, with comparable levels of artemisinin to the 100 mg of pure artemisinin in capsule resulted in much more rapid uptake and what appears to be significantly higher absorption and exposure to artemisinin, but this cannot be definitively proven given that these were not head-to-head studies. The authors conclude that bioavailability is similar between the tea and 500 mg artemisinin capsules, but that serum concentrations achieved are not sufficient for clinical efficacy. Of course, the results of clinical trials on this exact tea do not support this conclusion, because even at these relatively low serum levels, clinical efficacy was quite good in patients with uncomplicated acute malaria.
If one assumes that artemisinin is the only relevant compound in the whole tea, then these pharmacokinetic studies can perhaps only be interpreted as providing insufficient levels of artemisinin for clinical efficacy as these authors did. Given that the clinical trials show efficacy despite this, more evidence is provided for the theory that there is synergy among artemisinin and non-antimalarial components of the herb. If non-artemisinin components in the tea such as the methoxylated flavonoids are reducing resistance of the parasites to the artemisinin, then the serum levels of the artemisinin will not need to be as high to have good clinical efficacy compared with artemisinin in isolation. This theory requires study in formal trials to determine if it is true.
Artemisinin autoinduces its own catabolism, primarily in the gut wall, but also in the liver. CYP3A4, 2B6, and 2C19 are the main enzymes involved. CYP2B6 and 2C19 are believed to be autoinduced significantly by artemisinin. Over 7–10 days of ongoing administration of artemisinin, blood levels fall dramatically in healthy volunteers and patients with uncomplicated malaria. This effect is most pronounced in women. Whether this occurs with whole A. annua extract needs to be studied urgently. Until it has been, it should be assumed continuous use of the whole herb for more than 1 week would lead to a loss of efficacy for optimal patient safety.
Intravenous (IV) administration of artemisinins bypasses this problem but is rarely available in most malarious areas due to the cost of the agents and equipment. This creates the additional hurdle of needing to provide a sterile environment for administration of the drug which is often not practical in malaria-afflicted areas. However, IV artemisinins are equally effective to IV quinine and safer for treatment of cerebral malaria, and therefore there is a need to work to make this more available in hospitals in malarious areas.
Similar results have been noted empirically using the whole herb. Due to the first-pass metabolism in the liver, oral bioavailability of artemisinin is only 32%. However, one rodent study found that a whole plant concentrated extract of A. annua led to 40 times greater serum levels of artemisinin than when artemisinin alone was given orally to mice infected with Plasmodium chabaudi. Not surprisingly the whole-plant extract was far more effective at reducing parasitemia than purified artemisinin.
Taking grapefruit juice (350 mL double-strength fresh frozen juice, a known CYP3A4 inhibitor) daily for 5 days with 100 mg artemether increased oral bioavailability of the drug at first, but did not stop the time-dependent decrease in blood levels after ingestion in healthy volunteers. This supports the idea that while CYP3A4 is involved in catabolizing artemisinins in the gut wall, it is not induced significantly by repeated intake of artemisinins.
Given this information, it seems pointless to take A. annua or artemisinin for more than 7 days. It further highlights the futility of attempting to use either one for prevention of malaria. Anecdotal reports of people avoiding malaria infection while taking A. annua or artemisinin extracts are more likely due to mosquito bite prevention or simple luck.
SAFETY:
There are rare case reports of neurotoxicity associated with treatments with artemisinins, but such effects are also seen in some malaria patients without artemisinins, so cause-effect relationships with neurotoxicity have not been established. If artemisinins do cause neurotoxicity, the incidence is quite low. Artemisinins are safe and effective in treating malaria in pregnant women, at least during the second and third trimesters.
Nausea can occur with administration of the crude herb or extracts thereof, likely related to its intense bitterness. In fact, the only adverse effect noted in the clinical trials on A. annua tea has been nausea which remits when therapy is completed. Co-administration with common, mild anti-emetic herbs such as mint, ginger or other aromatic spices may alleviate or prevent nausea in some patients based on empirical evidence. The safety of A. annua in pregnancy has not been studied.
COUNTERFEIT DRUGS AND OTHER ISSUES:
With the rising popularity of artemisinins for treatment of malaria and the ongoing poverty in malaria-stricken parts of the world, there has been a rise in counterfeit artemisinins for sale. This parallels what has been seen with every antimalarial drug that becomes available. However, the high cost of artemisinins may be driving higher levels of counterfeiting as more profit can be made. Such behavior may be economically rational from the point of view of the person doing the selling, but is extremely harmful from the larger social perspective due to the multiple levels of negative effects on public health that result.
Ultimately the solution to counterfeit drugs is to alleviate poverty. Given the unlikeness of this any time soon, other solutions include creating cheaper artemisinin by microbial synthesis, creating better cultivars of A. annua, better policing of antimalarial drug manufacturers, reducing corruption in malaria-affected nations, and securing the evidence base for plant-based malaria therapies so that people can grow their own medicines.
CONCLUSION
The story of A. annua is instructive in many ways and offers hope as a multifaceted medicine. The whole herb became a source for an isolated compound (artemisinin or qinghaosu) which then became the template for multiple semisynthetic compounds. Though initially expensive and slow to become utilized, they have since become the dominant form of treatment for malaria worldwide. Despite the rapid awareness that they must be used in combination to avoid recrudescence and resistance, monotherapy continues and is resulting in loss of efficacy of this valuable therapy. Counterfeit drugs are worsening the problem by delaying actual therapy, causing patients to waste their already limited financial resources on useless medicines, and creating confusion and doubt in people’s minds about how and where to obtain actual drugs.
Whole-herb A. annua offers several potential ways out of these problems. First, it can be grown locally in malarious areas, allowing for a sustainable and self-supporting situation for communities afflicted with this disease. Second, it is effective despite relatively low levels of artemisinin, at least in part due to the resistance-inhibiting effects of methoxylated flavonoids in the whole plant. Third, it is cheap and safe. Fourth, it is potentially active against multiple tropical infectious diseases and may help with other diseases, including autoimmune conditions. Fifth, it may help control mosquito vectors. However, like isolated artemisinins, it is not effective at preventing recrudescence and requires combination therapy. What is unknown is whether it can be paired with other effective herbal antimalarials, several of which have been reported, and thus create effective, entirely local herbal remedies for malaria.
DISCLOSURE OF INTERESTS
Dr. J.E. Yarnell reports he is part-owner and president of Heron Botanicals, outside the submitted work.
REFERENCES
- Hsu E. The history of qing hao in the Chinese materia medica.
- Dhingra V, Rao KV, Narasu ML. Artemisinin: present status and perspectives.
- Wallaart TE, Pras N, Quax WJ. Seasonal variations of artemisinin and its biosynthetic precursors in tetraploid Artemisia annua plants compared with diploid wild-type.
- Baraldi R, Isacchi B, Predieri S, et al. Distribution of artemisinin and bioactive flavonoids from Artemisia annua L during plant growth.
- Luo SD, Ning BM, Hu WY, Xie JL. Studies on peroxides of Artemisia lancea.
- Liersch R, Soicke H, Stehr C, Tüllner HU. Formation of artemisinin in Artemisia annua during one vegetation period.
- Hsu E. Reflections on the ‘discovery’ of the antimalarial qinghao.
- Woerdenbag HJ, Lugt CB, Pras N. Artemisiaannua L: a source of novel antimalarial drugs.
- Meshnick SR. Artemisinin: mechanisms of action, resistance and toxicity.
- Shah NK, Schapira A, Juliano JJ, et al. Nonrandomized controlled trial of artesunate plus sulfadoxine-pyrimethamine with or without primaquine for preventing posttreatment circulation of Plasmodium falciparum gametocytes.
- Eziefula AC, Staedke SG, Yeung S, et al. Study protocol for a randomised controlled double-blinded trial of the dose-dependent efficacy and safety of primaquine for clearance of gametocytes in children with uncomplicated falciparum malaria in Uganda.
- Arango EM, Upegui YA, Carmona-Fonseca J. Efficacy of different primaquine-based antimalarial regimens against Plasmodium falciparumgametocytemia.
- Weed S. Healing wise…amazing artemisias. Wise Woman Ezine, 2005.http://www.susunweed.com/herbal_ezine/November05/healingwise.htm [accessed on 15 Dec 2013] .
- Liu KCS, Yang SY, Roberts MF, et al. The contribution of flavonoids to the antimalarial activity of Artemisiaannua.
- Elford BC, Roberts MF, Phillipson JD, Wilson RJ. Potentiation of the antimalarial activity of qinghaosu by methoxylated flavones.
- Weathers PJ, Towler MJ. The flavonoids casticin and artemetin are poorly extracted and are unstable in an Artemisia annua tea infusion.
- Stermitz FR, Scriven LN, Tegos G, Lewis K. Two flavonols from Artemisa annua which potentiate the activity of berberine and norfloxacin against a resistant strain of Staphylococcus aureus.
- Kumar S, Gupta AK, Pal Y, Dwivedi SK. In-vivo therapeutic efficacy trial with artemisinin derivative, buparvaquone and imidocarb diproprionate against Babesia equi infection in donkeys.
- Ćavar S, Maksimović M, Vidic D, Parić A. Chemical composition and antioxidant and antimicrobial activity of essential oil of Artemisia annua L from Bosnia.
- de Almeida GF, Horsted K, Thamsborg SM, et al. Use of Artemisia annua as a natural coccidiostat in free-range broilers and its effects on infection dynamics and performance.
- Lubbe A, Seibert I, Klimkait T, et al. Ethnopharmacology in overdrive: the remarkable anti-HIV activity of Artemisia annua.
- Islamuddin M, Farooque A, Dwarakanath BS, et al. Extracts of Artemisia annua leaves and seeds mediate programmed cell death inLeishmania donovani.
- Miller MJ, Walz AJ, Zhu H, et al. Design, synthesis, and study of a mycobactin-artemisinin conjugate that has selective and potent activity against tuberculosis and malaria.
- Utzinger J, N’Goran EK, N’Dri A, et al. Oral artemether for prevention of Schistosoma mansoni infection: randomised controlled trial.
- Zhang YQ, Ding W, Zhao ZM, et al. Studies on acaricidal bioactivities of Artemisia annua L extracts against Tetranychus cinnabarinus Bois (Acari: Tetranychidae).
- Ali Khan MMA, Jain DC, Bhakuni RS, et al. Occurrence of some antiviral sterols in Artemisia annua.
- Carrijo de Oliveira T, Oliveira Silva DA, Rostkowska C, et al. Toxoplasma gondii: Effects of Artemisia annua L on susceptibility to infection in experimental models In vitro and in vivo.
- Nibret E, Wink M. Volatile components of four Ethiopian Artemisia species extracts and their in vitro antitrypanosomal and cytotoxic activities.
- Ho WE, Peh HY, Chan TK, Wong WS. Artemisinins: Pharmacological actions beyond anti-malarial.
- Melillo de Magalhães P, Dupont I, Hendrickx A, et al. Anti-inflammatory effect and modulation of cytochrome P450 activities by Artemisia annuatea infusions in human intestinal Caco-2 cells.
- Zhao YG, Wang Y, Guo Z, et al. Dihydroartemisinin ameliorates inflammatory disease by its reciprocal effects on Th and regulatory T cell function via modulating the mammalian target of rapamycin pathway.
- Ryu JC, Park SM, Hwangbo M, et al. Methanol extract of Artemisia apiacea Hance attenuates the expression of inflammatory mediators via NF- κ B inactivation.
- Koo HN, Hong SH, Jeong HJ, et al. Inhibitory effect of Artemisia capillaris on ethanol-induced cytokines (TNF-alpha, IL-1alpha) secretion in Hep G2 cells.
- McIntosh HM, Olliaro P. Artemisinin derivatives for treating uncomplicated malaria.
- Mueller MS, Karhangomba IB, Hirt HM, Wemakor E. The potential of Artemisia annua L as a locally produced remedy for malaria in the tropics: agricultural, chemical and clinical aspects.
- De Donno A, Grassi T, Idolo A, et al. First-time comparison of the In vitro antimalarial activity of Artemisia annua herbal tea and artemisinin.
- Mueller MS, Runyambo N, Wagner I, et al. Randomized controlled trial of a traditional preparation of Artemisia annua L. (annual wormwood) in the treatment of malaria.
- WHO.
- Blanke CH, Naisabha GB, Balema MB, et al. Herba Artemisiae annuae tea preparation compared to sulfadoxine-pyrimethamine in the treatment of uncomplicated falciparum malaria in adults: a randomized double-blind clinical trial.
- Rasoanaivo P, Wright CW, Willcox ML, Gilbert B. Whole plant extracts versus single compounds for the treatment of malaria: synergy and positive interactions.
- Cheah SX, Tay JW, Chan LK, Jaal Z. Larvicidal, oviposition, and ovicidal effects of Artemisia annua (Asterales: Asteraceae) against Aedes aegypti, Anopheles sinensis, and Culex quinquefasciatus (Diptera: Culicidae).
- Sharma P, Mohan L, Srivastava CN. Growth inhibitory nature of Artemisia annua extract against Culex quinquefasciatus (Say).
- Bartarya R, Srivastava A, Tonk S, et al. Larvicidal activity of Artemisia annua L callus culture against Anopheles stephensi larvae.
- Tonk S, Bartarya R, Maharaj Kumari K, et al. Effective method for extraction of larvicidal component from leaves of Azadirachta indica andArtemisia annua Linn.
- Ramazani A, Sardari S, Zakeri S, Vaziri B. In vitro antiplasmodial and phytochemical study of five Artemisia species from Iran and in vivoactivity of two species.
- Zafar MM, Hamdard ME, Hameed A. Screening of Artemisia absinthium for antimalarial effects on Plasmodium berghei in mice: a preliminary report.
- Gathirwa JW, Rukunga GM, Njagi ENM, et al. In vitro anti-plasmodial and in vivo anti-malarial activity of some plants traditionally used for the treatment of malaria by the Meru community in Kenya.
- Kraft C, Jenett-Siems K, Siems K, et al. In vitro antiplasmodial evaluation of medicinal plants from Zimbabwe.
- Ene AC, Atawodi SE, Ameh DA, et al. Bioassay-guided fractionation and in vivo antiplasmodial effect of fractions of chloroform extract ofArtemisia maciverae Linn.
- Dua VK, Verma G, Agarwal DD, et al. Antiprotozoal activities of traditional medicinal plants from the Garhwal region of North West Himalaya, India.
- Nahrevanian H, Sheykhkanlooye Milan B, Kazemi M, et al. Antimalarial effects of Iranian flora Artemisia sieberi on Plasmodium berghei in vivoin mice and phytochemistry analysis of its herbal extracts.
- Taherkhani M, Rustaiyan A, Nahrevanian H, et al. Comparison of antimalarial activity of Artemisia turanica extract with current drugs in vivo.
- Karunamoorthi K, Sabesan S, Jegajeevanram K, Vijayalakshmi J. Role of traditional antimalarial plants in the battle against the global malaria burden.
- Challand S, Willcox M. A clinical trial of the traditional medicine Vernonia amygdalina in the treatment of uncomplicated malaria.
- Willcox ML, Graz B, Falquet J, et al. Argemone mexicana decoction for the treatment of uncomplicated falciparum malaria.
- Benoit-Vical F, Valentin A, Da B, et al. N’Dribala (Cochlospermum planchonii) versus chloroquine for treatment of uncomplicated Plasmodium falciparum malaria.
- Ankrah NA, Kyarko AK, Addo PGA, et al. Evaluation of efficacy and safety of a herbal medicine used for the treatment of malaria.
- Bunnag D, Harinasuta T, Looareesuwan S, et al. A combination of quinine, quinidine and cinchonine (LA 40221) in the treatment of chloroquine resistant falciparum malaria in Thailand: Two double-blind trials.
- Yarnell E, Abascal K. Botanical treatment and prevention of malaria. Part 2–Selected botanicals.
- Udeinya IJ, Brown N, Shu EN, et al. Fractions of an antimalarial neem-leaf extract have activities superior to chloroquine, and are gametocytocidal.
- Isah AB, Ibrahim YK, Iwalewa EO. Evaluation of the antimalarial properties and standardization of tablets of Azadirachta indica (Meliaceae) in mice.
- Becky Andrews, ND, personal communication , 2006.
- Udeinya IJ, Mbah AU, Chijioke CP, Shu EN. An antimalarial extract from neem leaves is antiretroviral.
- Sharma SK, Dua VK, Sharma VP. Field studies on the mosquito repellent action of neem oil.
- Luong K, Dunkel FV, Coulibaly K, Beckage NE. Potential use of neem leaf slurry as a sustainable dry season management strategy to control the malaria vector Anopheles gambiae (DIPTERA: CULICIDAE) in west African villages.
- Nandakumar DN, Nagaraj VA, Vathsala PG, et al. Curcumin-artemisinin combination therapy for malaria.
- Isacchi B, Bergonzi MC, Grazioso M, et al. Artemisinin and artemisinin plus curcumin liposomal formulations: enhanced antimalarial efficacy against Plasmodium berghei-infected mice.
- Aditya NP, Chimote G, Gunalan K, et al. Curcuminoids-loaded liposomes in combination with arteether protects against Plasmodium bergheiinfection in mice.
- Vathsala PG, Dende C, Nagaraj VA, et al. Curcumin-arteether combination therapy of Plasmodium berghei-infected mice prevents recrudescence through immunomodulation.
- Martinelli A, Rodrigues LA, Cravo P. Plasmodium chabaudi: efficacy of artemisinin + curcumin combination treatment on a clone selected for artemisinin resistance in mice.
- Ajayi NA, Ukwaja KN. Possible artemisinin-based combination therapy-resistant malaria in Nigeria: a report of three cases.
- Cheng Q, Kyle DE, Gatton ML. Artemisinin resistance in Plasmodium falciparum: A process linked to dormancy?
- Lim P, Dek D, Try V, et al. Ex vivo susceptibility of Plasmodium falciparum to antimalarial drugs in western, northern, and eastern Cambodia, 2011–2012: association with molecular markers.
- Hübner AT. Treatment with Hypericum perforatum L does not trigger decreased resistance in Staphylococcus aureus against antibiotics and hyperforin.
- Nelson RRS. Selection of resistance to the essential oil of Melaleuca alternifolia in Staphylococcus aureus.
- Bonati M, Severino F, Bagnati R, et al. Millet-porridge with Artemisia annua as first aid for African children with malaria?
- de Ridder S, van der Kooy F, Verpoorte R. Artemisia annua as a self-reliant treatment for malaria in developing countries.
- Zeng Q, Qiu F, Yuan L. Production of artemisinin by genetically-modified microbes.
- Räth K, Taxis K, Walz G, et al. Pharmacokinetic study of artemisinin after oral intake of a traditional preparation of Artemisia annua L (annual wormwood).
- Gordi T, Hai TN, Hoai NM, et al. Use of saliva and capillary blood samples as substitutes for venous blood sampling in pharmacokinetic investigations of artemisinin.
- Simonsson USH, Jansson B, Hai TN, et al. Artemisinin autoinduction is caused by involvement of cytochrome P450 2B6 but not 2C9.
- Ashton M, Nguyen DS, Nguyen VH, et al. Artemisinin kinetics and dynamics during oral and rectal treatment of uncomplicated malaria.
- Ashton M, Hai TN, Sy ND, et al. Artemisinin pharmacokinetics is timedependent during repeated oral administration in healthy male adults.
- Kyu HH, Fernández E. Artemisinin derivatives versus quinine for cerebral malaria in African children: a systematic review.
- Meshnick SR, Taylor TE, Kamchonwongpaisan S. Artemisinin and the antimalarial endoperoxides: From herbal remedy to targeted chemotherapy.
- Titulaer HA, Zuidema J, Kager PA, et al. The pharmacokinetics of artemisinin after oral, intramuscular and rectal administration to volunteers.
- Elfawal MA, Towler MJ, Reich NG, et al. Dried whole plant Artemisia annua as an antimalarial therapy.
- van Agtmael MA, Gupta V, van der Graaf CA, van Boxtel CJ. The effect of grapefruit juice on the time-dependent decline of artemether plasma levels in healthy subjects.
- McGready R, Cho T, Cho JJ, et al. Artemisinin derivatives in the treatment of falciparum malaria in pregnancy.
- Newton PN, Fernández FM, Plançon A, et al. A collaborative epidemiological investigation into the criminal fake artesunate trade in South East Asia.
- Dondorp AM, Newton PN, Mayxay M, et al. Fake antimalarials in Southeast Asia are a major impediment to malaria control: multinational cross-sectional survey on the prevalence of fake antimalarials.
- Paul S, Khanuja SP, Shasany AK, et al. Enhancement of artemisinin content through four cycles of recurrent selection with relation to heritability, correlation and molecular marker in Artemisia annua L.
- Delabays N, Simonnet X, Gaudin M. The genetics of artemisinin content in Artemisia annua L and the breeding of high yielding cultivars.