Abstract
Women experience acute myocardial infarctions (AMI) 10 years later than men – evidence that estrogen is protective is not consistent. Ovulatory disturbances (low progesterone but normal estradiol levels) silently occur in >33% of all cycles. Progesterone-based (cycle-timed serum or saliva) levels or urinary metabolite excretions are necessary to diagnose silent ovulatory disturbances within regular, normal length menstrual cycles. Progesterone acts biphasically in vitro – initial proliferation changes to differentiation. It also suppresses or complements estradiol’s actions. Basic and clinical studies show that progesterone is positively related to endothelial function/blood flow, influences vascular smooth muscle cells and cardiac electrical signals. Several studies in primates document that high fat-fed subordinate females have higher stress, fewer ovulatory cycles and lower progesterone levels but similar cycle lengths as menstruating, dominant females. Subordinate females develop arterial plaque similar to high fat-fed males. In population-based prospective data, women with early pregnancy progesterone deficiency, stress and multiple miscarriages are at five-fold higher AMI risk. Also, early menopausal AMI are associated with significantly more pre-/perimenopausal anovulatory cycles. Given the multi-dimensional positive effects of progesterone on the cardiovascular system (CVS), and persuasive data from primate and human studies associating increased AMI with ovulatory disturbances, this review presents a new CVS protection hypothesis for women – ovulatory cycles with balanced progesterone-estradiol levels decrease women’s risks for AMI. Based on this new theory, it is believed that progesterone as well as estradiol is required during the premenopausal years to prevent women’s early heart disease.
INTRODUCTION
It is well established that women having their first heart attack (acute myocardial infarction, AMI) are approximately 10 years older than men. Although the risk for, and consequences of, coronary heart disease (CHD), AMI and other cardiovascular diseases (CVD, including stroke, heart failure and peripheral vascular diseases) are similar between genders, factors involved may be different. These issues have recently been comprehensively reviewed and guidelines for women’s CVD prevention are available. Although overall, women’s CHD incidence and mortality are lower than men’s throughout the lifecycle, cardiovascular diseases (heart diseases and stroke) are the largest single reason for death in women of all ages. CHD remains a current and urgent issue for women due to recent British Columbia-wide data showing increasing AMI mortality rates in younger women (defined as those ≤55 years old). In other age cohorts and in men, AMI mortality rates have seen decreases over time.
Furthermore, there may be gender differences in the effectiveness of CHD prevention strategies. For example, statin treatment is prescribed and effective for secondary prevention in both men and women but only in 2013 did a meta-analysis suggest that statins are effective for primaryprevention in women.7 Statin use for women’s primary CVD prevention continues to lack data in large cohorts on its cost-effectiveness, side-effect profiles and number needed to treat. Low dose aspirin has been reported to decrease men’s risk of first AMI but does not in women; aspirin does, however, decrease stroke in hypertensive women aged ≥65. The Framingham study showed that the presence of diabetes attenuates women’s “cardiovascular protection” and is related to increased CHD risks and higher mortality. However, impaired fasting glucose also increases women’s but not men’s CHD risk, further demonstrating gender-related complexities in the pathophysiology of CVD.
For many years, estrogen has been understood to provide women with “cardiovascular protection” – a corollary is that “postmenopausal estrogen deficiency” causes heart disease. These ideas were based on observational data in large cohorts such as the Nurse’s Health Study. These studies have been criticized, however, because estrogen-treated women were healthier, often had better access to health care and only those treated women without side effects continued estrogen therapy. However, beginning in 2002, two large estrogen or estrogen-progestin women’s primary prevention randomized controlled trials (RCTs) showed negative cardiovascular results. More recently, abstract-presented early results in younger menopausal women in the Kronos Early Estrogen Prevention Study showed that neither transdermal estradiol nor oral conjugated estrogen (each with lower dose cyclic progesterone) prevented progression of coronary artery calcification or carotid intima media thickness. Further, as early as 1997, in the Ancel Keys lecture, epidemiology physician Barrett-Connor while asserting that women’s cardiovascular system was “superior” to that of men, stated: “Plasma estrogen levels do not explain CHD in either sex; . . . new hypotheses are needed”.
Several recent publications from the Centre for Menstrual Cycle and Ovulation Research (CeMCOR) in addition to the existing body of literature on menstrual cycles and ovulation have led to the hypothesis that cardiovascular protection in women is related to the progesterone as well as the estradiol within normally ovulatory pre/perimenopausal menstrual cycles. These new observations are: oral micronized progesterone in a 3-month randomized placebo-controlled RCT in healthy menopausal women tends to improve endothelial function (15% increase in flow but non-significant) and showed cardiovascular safety. A recent meta-analysis of prospective studies documented that undetected, clinically silent “ovulatory disturbances,” meaning anovulation or short luteal phases within regular menstrual cycles, occurred almost 50% of the time; ovulatory disturbances were significantly related to negative bone changes. As well, a whole population study of ovulation within one cycle in over 3000 regularly cycling women showed that anovulation (diagnosed by a cycle-timed serum progesterone level) occurred about a third of the time. Together these data showed that oral micronized progesterone has short term beneficial effects and safety; silent progesterone deficiency (ovulatory disturbances) is common despite sufficient estradiol in regular cycles.
Thus, the primary purpose of this evidence-based review is to propose a new women’s heart protection hypothesis: normal progesterone levels in ovulatory women counterbalance or complement estradiol’s actions leading to subsequent protection from early CHD.
ESTROGEN AND CARDIOVASCULAR DISEASE
The notion persists that endogenous estradiol levels are the reason that women’s AMI incidence lags behind men’s. Further, in assessment of women’s cardiovascular risks, estrogen deficiency is considered to be the key without considering that a woman with amenorrhea is also progesterone deficient. In addition, it is assumed that if women have regular cycles of normal lengths (21–35 days), they are hormonally normal and therefore heart disease is not a worry. Menopausal women, who are defined in epidemiology as being 1 year after their final menstruation, are believed to have “estrogen deficiency,” although low levels of both progesterone and estradiol are normal in menopausal women. Despite the unsuccessful outcomes in large, long-duration RCT estrogen or estrogen-progestin therapy primary and secondary CVD prevention studies in menopausal women and of higher dose estrogen therapy in men, this “estrogen-heart protection” notion for women persists.
At the core in explanation of the new progesterone-estradiol premenopausal balance hypothesis for CVD protection is the assumption that regular cycles are inevitably ovulatory as vigorously asserted by some. Other experts declare that the high levels of premenopausal progesterone within ovulatory menstrual cycles are a “unique disorder”. Despite these assertions, normal ovulation is by no means inevitable in clinically normal menstrual cycles – multiple researchers using various assessment methods have documented lack of ovulation and/or low progesterone levels occurring despite regular cycles. Furthermore, estradiol levels have been reported to be similar in anovulatory and ovulatory cycles.
PHYSIOLOGY OF MENSTRUATION:
Due to the varying methodologies reported in the literature for the assessment of ovulation within menstrual cycles, it is important to understand the reliability and relevance of the various detection methods. The gold standard for ovulation documentation is real-time surgical visualization of an egg being extruded from the rupturing follicle (see http://youtu.be/KjuJr5i1OGk). Thus all ovulation detection methods are indirect; most either measure progesterone or some action of progesterone (as in increasing core temperature). Common validated methods include cycle-timed single or multiple serum or salivary progesterone levels, a three-fold follicular to luteal increase in urinary excretion of the progesterone metabolite, pregnanediol glucuronide (PdG) or quantitative elevation of the first morning temperature [quantitative basal temperature (QBT)]. Although the mid-cycle luteinizing hormone (LH) peak usually precedes and triggers ovulation, an LH peak may occur without subsequent ovulation – it has sensitivity without specificity.
Anovulation means failure to release an egg; it is the post-ovulatory corpus luteum that produces progesterone, therefore progesterone levels remain low throughout an anovulatory menstrual cycle. As some clinicians use the term “anovulation” to mean oligo- or amenorrhea, for purposes of this article, the use of the term “anovulation” describes a menstrual cycle without progesterone levels achieving ≥9.54 nmol/L for at least 3 days during the cycle.
Menstrual cycles are timed from the first day of flow, thus cycle day 10 is 10 days after the onset of menstruation. Menstrual cycle lengths (from the first day of flow until the day before the next flow) are accepted as normal if between 21 and 35 days in length and with usual variability of less than 7 days. Menstrual cycle lengths roughly indicate integrated cycle estradiol levels – those that are shorter tend to have higher estradiol levels and those 36 days or longer (oligomenorrhea) tend to have lower estradiol levels. Amenorrhea, meaning no flow for three or more cycles, indicates that low levels of both estradiol and progesterone are present unless there is a pregnancy or its occurrence is in a woman with anovulatory androgen excess, commonly called polycystic ovary syndrome (PCOS). In PCOS, even with amenorrhea, estradiol levels are tonically high. (This review will not further discuss PCOS since this syndrome involves only about 5% of the premenopausal population, is complicated by androgen excess and often associated with obesity and insulin resistance as well as major psychosocial stress; all of these individually increase CVD risks.)
“Ovulatory disturbances” is a generic term for decreased progesterone production within regular cycles. It is defined as clinically normal menstrual cycles (regular, and of normal length) that are either anovulatory or are ovulatory but having a shortened luteal phase length; short luteal phases are the more common of ovulatory disturbances. There are no large prospective population-based studies (i.e. whole cohort or random sample of a discrete group and to include a participation rate of ideally more than 50%) that have reported ovulatory disturbances, but prospective studies suggest that it is relatively common. Furthermore, based on a cross-sectional single cycle-timed serum progesterone measurement within regular cycles in a population-based cohort of over 3500 spontaneously menstruating Norwegian women aged 20–49.9 years, at least 30% of all regular, normal-length cycles are anovulatory.
THE ROLE OF PROGESTERONE IN CARDIAC PHYSIOLOGY
Progesterone is women’s post-ovulatory or luteal phase menstrual cycle hormone, in contrast to estradiol, menstrual cycle’s follicular phase hormone. The absolute and relative amounts of the two steroids produced during the menstrual cycle must be noted since this relationship is obscured by the fact that both hormones are commonly presented graphically (for example, many are readily available in “Google Images” that are without units) (Figure 1a). In reality, estradiol is reported in “pmols” while progesterone is reported in “nmols”. To show estradiol-progesterone menstrual cycle relationships, each hormone can be reported as percentage changes related to its level on cycle day 1; in the normal ovulatory menstrual cycle, estradiol rises to a peak that is 240% above its low during the follicular phase – by contrast progesterone rises 1400% above its follicular phase baseline (Figure 1b).
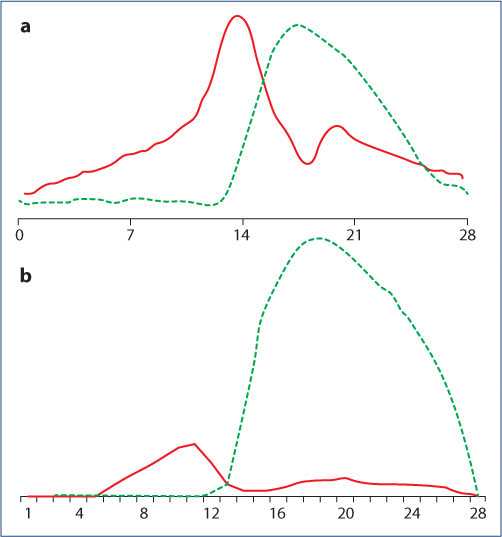
Figure 1:This two-part figure shows in a) an internet-extracted diagram of the menstrual cycle hormones, estradiol and progesterone plotted across menstrual cycle days without any values for the hormone levels (www.thenakedscientists.com and adapted by JC Prior 5/2009). Estradiol is shown having similar levels as progesterone. The Y axis has no units—estradiol is in solid red line and progesterone is the dashed green line. By contrast, in b) the percentage changes of estradiol and progesterone from their respective low follicular phase baselines are shown (Y axis) illustrating the much greater increase in progesterone (dashed green line) than estradiol (solid red line) levels during the menstrual cycle. This is adapted by JC Prior from Nielsen et al. J Clin Endocrinol Metab. 1990;70:1431–7.
PROGESTERONE’S CELLULAR AND SUBCELLULAR CARDIOVASCULAR-RELATED ACTIONS:
In vitro studies of cells in culture are typically short-term experiments lasting only a few days. These studies demonstrate that both progesterone and estradiol cause cell growth or proliferation – this is true in different tissues and in differing animal species. However, if the culture time is extended, then progesterone and estradiol have differing actions – progesterone is biphasic with initial proliferation followed by anti-proliferative and differentiation/maturation effects. Thus in both human biology (for example, the importance of adequate luteal phase length for fertility) and in in vitro studies, progesterone’s duration of action is important.
STEROID RECEPTORS:
It is known that steroid receptors show promiscuous interactions, but the receptor-mediated actions of estradiol and progesterone also indicate specific and extensive cross-talk. Estradiol, in general, leads to up-regulation of progesterone receptors. This does not mean that when estradiol levels are low, however, progesterone is inactive. Each tissue may have a different steroid action threshold; for example, bedtime oral micronized progesterone therapy, in doses keeping the blood level above the luteal phase threshold for 24 hours, has been documented in an RCT to be effective in decreasing hot flushes/flashes and night sweats in menopausal women despite their known normally low estradiol levels. Progesterone also causes inactivation of estradiol receptors and enzymatic changes leading to decreased estradiol actions. Further, progesterone has important counterbalancing interactions with cortisol. In osteoblasts, for example, cortisol inhibits bone formation while progesterone counterbalances this, and through specific progesterone-receptor mediated actions, directly promotes osteoblastic bone formation.
MYOCARDIAL OR VASCULAR CELLS:
Progesterone cardiovascular physiology as it relates to myocardial or vascular cells, endothelial function with direct stimulation and indirect vasodilatation through nitric oxide production, related to angiogenic factors, responses to adrenergic stimulae or to electrical responses of the cardiovascular system have been previously reported on, thus are only briefly summarized in this review. It is postulated that fibroblast and myocardial cell overgrowth are induced by cardiac injury and high blood pressure, in turn leading to cardiovascular disease and hypertension. An in vitro study of myocardial fibroblasts showed that 17b estradiol and progesterone (but not estrone nor estriol) inhibited fibroblast proliferation. However, hormone exposure time in culture was limited to 24 hours. Thus, given the biphasic cellular actions of progesterone,? with longer exposure it is plausible that progesterone may induce fibroblast differentiation and regeneration thus aiding in cardiac injury repair and maintenance (similar to the progesterone’s activity in bone formation). Both estradiol and progesterone after three days in culture similarly inhibited mitogen-stimulated proliferation of human umbilical vein smooth muscle cells. Likewise, with longer term progesterone culture, vascular smooth muscle cells might show differentiation.
ENDOTHELIAL FUNCTION AND THE NITRIC OXIDE SYSTEM:
Blood vessels can be dilated directly by vascular smooth muscle actions or indirectly through nitric oxide produced in endothelial cells. Endothelial progenitor cells that arise in the bone marrow and hone in on tissues as needed, are understood to be fundamental to cardiovascular physiology. Several in vitro studies have shown that premenopausal women’s circulating endothelial progenitor cells are more effective at colony formation, and respond better to oxidative stresses than in men of a similar age. Circulating endothelial progenitor cells have been shown to increase in number at the LH peak and in the mid-luteal phase in 11 progesterone-documented ovulatory premenopausal women. However, endothelial cell number correlated with estradiol, but not with progesterone levels. Nitrites and endothelial nitric oxide synthetase (eNOS) are stimulated by progesterone; this stimulation is similar to or greater than the effects of estradiol.
RESPONSES TO ADRENERGIC STIMULAE:
Catecholamines are important regulators of responses of the cardiovascular system. As such they are both important for essential functions such as maintaining adequate blood pressure during pregnancy, ensuring adequate upright blood pressure and protective responses during dehydration or volume depletion. However, vasopressors may also be harmful to the cardiovascular system, thus, modulation of responses to adrenergic stimulae are likely important in vascular biology. Progesterone actions on vascular responses have been studied both in vivo in rats and in vitro in vascular smooth muscle tissues (devoid of endothelial cells). These studies demonstrated that progesterone antagonizes vasoconstriction (stimulated by various agents including arginine vasopressin and norepinephrine) likely through calcium-channel effects. This was confirmed in humans in a randomized cross-over trial which showed that, with norepinephrine transport inhibition during a head-up tilt procedure, there were greater counter-regulatory responses in the hormonally confirmed luteal phase than during the follicular phase.
CARDIAC ELECTRICAL CONDUCTION SYSTEM EFFECTS:
Arrhythmias resulting from conduction disturbances within the heart are responsible for many strokes, some syncope and even sudden death. Recent evidence suggests there are hormonal influences on the cardiac electrical repolarization rate or the QT rate-corrected (QTc) interval. The long QT2 syndrome in women is associated with ventricular arrhythmias and sudden death. To clarify the relationship of sex hormones, prepubertal rabbits with a genetic long QTc defect were ovariectomised and then in a random fashion were treated with estradiol, progesterone, dihydrotestosterone or placebo and observed over eight weeks for cardiac arrhythmias or sudden cardiac death. Estradiol was found to be proarrhythmic and increased risk of sudden cardiac death versus placebo and all other treatments. In contrast, progesterone compared with both estradiol and placebo, decreased arrhythmias and risk of sudden cardiac death.
ENERGY METABOLISM:
Progesterone is known to increase core or basal temperature; interestingly, estradiol appears to counterbalance progesterone’s thermogenesis. The progesterone-temperature effect is now known to be effected through hypothalamic progesterone receptors. The physics of progesterone’s luteal phase small temperature increase (about 0.25°C) indicate that concomitant increased energy consumption is required for weight maintenance. This has been demonstrated in a prospective diet-monitored menstrual cycle study where it was found that weight-stable premenopausal women consumed about 300 kilocalories more per day if they were ovulatory versus anovulatory.
It has also been understood for decades that women’s reproductive function and energy availability are related. The Frisch hypothesis of body fat and age at menarche has been criticized but the fundamental observation is correct. Given these facts, progesterone may have interactions with food-seeking/appetite, insulin levels, energy intakes, brain appetite and satiety hormones and to other aspects of intermediary metabolism. Alternatively, anthropology studies show that increased energy expenditures (through exercise/work demands) are associated with lower progesterone levels. However, the lengths of the luteal phases were not considered. Given the higher energy intakes in the luteal phase, it is understandable that higher insulin levels are also observed in that cycle phase. An RCT in which 10 days of oral micronized progesterone was administered showed a tendency to decrease fasting insulin levels and improve markers of insulin resistance in young women with PCOS.
CARDIOVASCULAR EFFECTS OF PROGESTERONE IN PRIMATES
Cynomolgus monkeys have similar menstrual cycle hormones and cycle lengths as humans, and are therefore an appropriate model in which to study relationships of ovulation to cardiovascular diseases in women. In the 1980s, Bowman Gray School of Medicine at Wake Forest University imported young adult cynomolgus monkeys from Southeast Asia for studies of cardiovascular risk factors. At least three identical early experiments (each published separately without indication if there were overlapping data) fed male and female animals a “Western” style high fat diet for 30 months. The primary outcome was coronary artery and large vessel plaque and fatty streaks. The animals were housed in “family groups” that included a single vasectomized male and four or five females. In all experiments social interactions were documented by systematic observations. Researchers observed that a couple of females per group were groomed more, got to feed first and were consistently dominant in female social relationships. Conversely, three or more of the females were rather isolated, fed last and had less grooming – these dominant and subordinate social status designations were stable within a given animal over time. Along with social observations, the investigators trained the females to submit to daily vaginal swabbing to detect menstruation and to have blood samples for progesterone levels collected cycle day 7 and every three days starting cycle day 12. Animals were also weighed regularly and estradiol data were available for a few animals. For all animals, at necropsy, bilateral adrenal weight (adjusted for body weight) was measured. Thirty months of demographic, social behavioural, menstrual cycle, ovulatory and hormonal results are presented in Table 1. This experiment is presented as it is the only study in which number of cycles were reported. All three experiments, each involving about 23 females, included both dominant and subordinate females and reported peak progesterone levels and the proportion of anovulatory cycles over 30 months. In all experiments, those animals that were dominant had fewer anovulatory cycles (approximately 3–5%) versus the subordinate animals with anovulation rates of 16–25%. Furthermore, all coronary artery histology results indicated greater coronary atherosclerosis in the males and in subordinate females than in the dominant females. Results of coronary artery observations of one study are shown in Figure 2.
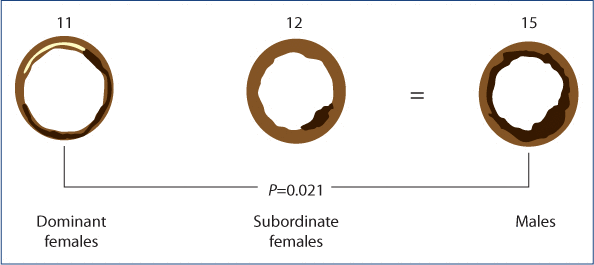
Figure 2:This diagram shows the coronary artery results of thirty months of eating a Western-style higher fat diet in cynomolgus monkeys. They were all housed in family groups of one vasectomized male with 4–6 females. Females differed in social status (dominant, subordinate) and their proportion of regular menstrual cycles with ovulatory disturbances – subordinate females had more ovulatory disturbances and similar vascular plaque burden as the males while the dominant, ovulatory females maintained normal coronary arteries. Numbers of animals are shown above. Drawn from data in Kaplan et al.

Table 1:Behavioral, physiological and morphological characteristics of reproductively intact dominant and subordinate female cynomolgus monkeys.
These cynomolgus monkey experiments consistently document that socially stressed animals having subordinate status have larger body weight-adjusted adrenal glands and are more likely to experience ovulatory disturbances within regular menstrual cycles. However, in one experiment three of the subordinate animals developed oligomenorrhea and, as expected, had lower estradiol levels. Given that these studies report on cycle lengths showing no difference between social status groups, and that all three studies showed that body weight was equal or very similar between groups, it can be inferred that these stressed and isolated monkeys, like women with ovulatory disturbances, usually have normal estradiol levels. Despite this assumption, it has been argued that the abnormal coronary arteries in subordinate animals were explained by “estrogen deficiency”. However, the data speak to the fact that the predominant perturbation of reproductive function was in ovulation (and thus lower progesterone) rather than in cycles (and thus lower estradiol). The first report of studies of this design summarized that “subordinate females had impaired ovarian function (increased frequency of anovulatory cycles and luteal phase deficiencies)”. Therefore, these data demonstrate a direct relationship between subclinical ovulatory disturbances and increased coronary artery disease in regularly menstruating female primates.
Additional primate data show that estradiol and progesterone both prevent coronary artery spasm acting through endothelial signals or through actions on vascular smooth muscle cells. Whether data support a direct positive cardiovascular effect of progesterone and negative consequences in its absence, or whether progesterone must be present to counterbalances primary negative heart effects of stress hormones cannot be determined from these studies. The subordinate female monkeys compared to dominant females had larger adrenal glands, and in one report showed higher ACTH-stimulated cortisol levels. However, the adrenal weights were positively related to the percentage of anovulatory cycles (r=0.43, P<0.05). That raises the question about whether cortisol has direct atherosclerotic effects. This question has been addressed in the discussion of endothelial function in humans.
PROGESTERONE, OVULATION AND THE CARDIOVASCULAR SYSTEM IN WOMEN
THE PREVALENCE AND INCIDENCE OF OVULATORY DISTURBANCES:
Based on a meta-analysis and emerging population data – disturbances of ovulation are common within clinically normal menstrual cycles in premenopausal women. This has been documented since the early 1980s. Eighty-two percent of women screened to be normally ovulatory in two consecutive cycles in a prospective 1-year study experienced at least one ovulatory disturbance. This study included premenopausal women of varying exercise habits, from “normally active” to those training for and running marathons. Women were non-smokers and did not use hormonal contraception, were between 20 and 42 years of age, with a normal body mass index (18.5–24.9 kg/m). Further, included women had no (overt) eating disorder and were not compulsive exercisers. Results showed that marathon-trained runners, despite lower body weights and body fat, were no more likely to experience ovulatory disturbances. Their cycle lengths remained within normal limits for all but about 1% of cycles that showed oligomenorrhea. The ovulatory characteristics of this cohort’s total menstrual cycles across that study year are shown in Figure 3 illustrating how common ovulatory disturbed cycles are, even in a homogenous cohort of healthy premenopausal women, and that short luteal phases are much more prevalent than anovulatory cycles.
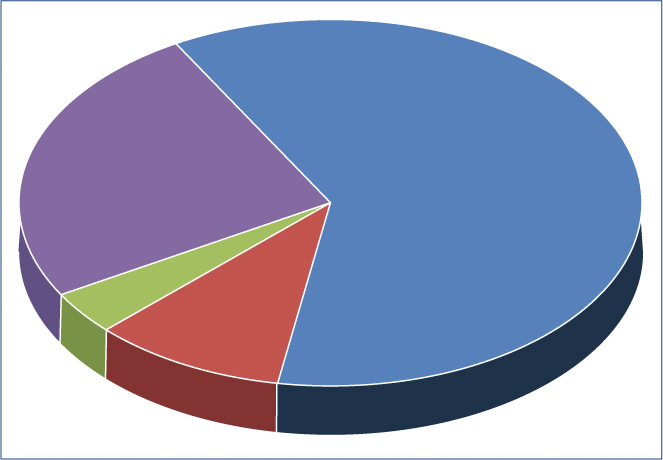
Figure 3:This pie chart describes the incident ovulatory disturbances within largely regular and normal length menstrual cycles (n=782): anovulation (4%)=green; short luteal phases (25%)=purple; normally ovulatory cycles (61%)=blue; and incomplete or could not analyze ovulation cycles (10%)=red. These data were from a prospective 1-year observational study in 66 initially proven-ovulatory, healthy premenopausal women. Redrawn from data in Prior et al.
Early reports on the adaption of sex organs in response to stress in addition to more recent studies, suggest that ovulatory disturbances represent an adaptation to a variety of stressors including unusual or new exercise-training. Progesterone is produced in very large quantities and this requires a major “effort” which would be spared in the distressed individual by opting to produce less progesterone while preserving estradiol levels and normal menstrual cycle-lengths. These reproductive adaptations have since been supported by an anthropological review of human reproduction in varying environments and seasons. Stressors that can influence ovulation may initially seem quite minor, as in cognitive dietary restraint (normal-weight women who worry about weight gain and obesity), but they are still associated with higher rates of ovulatory disturbances and, as in subordinate monkeys, related to higher cortisol exposures. Also, younger women, whose complex reproductive system is still maturing and developing normally ovulatory cycles (within 10–12 years of menarche), are more likely to respond to stressors with ovulatory disturbances.
Prospective studies of ovulation and menstrual cycles show that ovulatory disturbances are common. A recently published meta-analysis of prospective studies of menstrual cycles and ovulatory characteristics included six investigations in healthy premenopausal women. In these studies averaging 2 years’ duration in 436 individual women whose ages spanned the late teens to the early 40s, the median split of within-cohort percentage of ovulatory cycles was 31–39%. Looked at another way, despite normal mean cycle lengths averaging 27 days, a mean of 49% of women experienced one or more ovulatory disturbances per year (range 13–82%) (Table 2). Also, in a population-based whole-county cross-sectional study in Norway (participation rate 58.8%) using a cycle-timed progesterone threshold level of ≥9.54 nmol/L, the ovulation point prevalence was 56.3% to 65.8% depending on whether women provided investigators with information on the start date of only their last cycle or both their last and next cycles, respectively.

Table 2:Meta-analysis of prospective menstrual cycle and ovulation/luteal phase length studies.
It is noteworthy that women in perimenopause (the transition to menopause) may or may not menstruate regularly, and clearly have increased rates of ovulatory disturbances and infertility. Furthermore, perimenopausal women are also more likely than similar aged premenopausal women to experience chest pain and arrhythmias. Although the discussion of perimenopause and women’s cardiovascular risks is beyond the scope of this article, it is important to understand the increased cardiovascular risk in this population with ovulatory disturbances.
Thus, although perhaps previously unrecognized, silent ovulatory disturbances appear to be sufficiently common that they could theoretically relate to women’s risk of early heart attack.
REPRODUCTIVE VARIABLES RELATED TO CARDIOVASCULAR RISK:
In general the longer a woman’s reproductive lifespan (the number of years from a woman’s menarche to menopause) the lower her cardiovascular risk. In light of the proposed hypothesis, this ideally means longer exposure to both estradiol and progesterone. A number of other studies have examined heart attack risks related separately to age at menarche, age at menopause, nulliparity, parity, reproductive lifespan and cycle regularity or irregularity but none of these have documented ovulation or luteal phase length. However, it is known, based on population data, that women with an early age at menarche have greater likelihood of adult obesity which is a cardiovascular risk factor. Earlier menarche in population data is also associated with greater all-cause mortality. It is also reasonably clear that women with early menopause (generally before age 40) have an increased risk for heart attacks. Premenopausal bilateral ovariectomy has long been understood to put women at CVD risk but a 1-year comparative RCT of conjugated equine estrogen versus the synthetic medroxyprogesterone showed more weight gain and abnormal cardiovascular risks in women treated with the equine estrogen.
It is known that irregular menstrual cycles are associated with increased risks for heart attacks – irregular cycles are an indicator of lower estradiol levels (in women without PCOS) but also of lower progesterone levels associated with ovulatory disturbances that are more prevalent in irregularity. Currently however, there are no prospective data in women that have assessed menstrual cycle and ovulatory characteristics, cardiovascular risk factors, psychosocial stressors and documented subsequent AMI and CHD experiences.
Historically, before effective contraceptive strategies were available, nulliparity in married women was likely to indicate an increased difficulty conceiving – this subfertility was most commonly due to ovulatory disturbances. Now, women can choose whether or not to bear children. Data indicate that nulliparity is increased in women experiencing sudden cardiac death (RR=3.8) in a case-control study within married women. However, not all studies show a nulliparity relationship with women’s CVD risk perhaps because now it is usually related to a reproductive choice. Studies have shown that the risk relationship is U or J shaped, meaning that risk for CVD is increased in women who bear one child, lower in those bearing between two and four children, and again increases in women who bear five or more children. In parous women, the increased risk of CVD is partially attributed to history of pregnancy complications and dyslipidemia; however, it is clear other factors may also be involved that have not yet been identified.
INDIRECT, NITRIC OXIDE MEDIATED CONTROL OF VASCULAR ENDOTHELIUM AND BLOOD FLOW:
As previously discussed, endothelial function is fundamental to cardiovascular health. The indirect or nitric oxide-related increased blood flow response is triggered by temporary vessel occlusion and then release causing increased flow (“flow mediated dilatation”). As an experimental or clinical test, endothelial function may be assessed using ultrasound and post-occlusion changes in brachial artery diameter or it can be assessed more complexly and in smaller blood vessels using forearm strain gauges during venous occlusion plethysmography. Acetylcholine in standardized, increasing doses is infused locally via artery to stimulate the release of endogenous nitric oxide by endothelial cells.
Although there are few human investigations that have studied endothelial effects of progesterone separate from estrogen/estradiol, one study using venous occlusion plethysmography in 27 healthy early menopausal women during intra-arterial infusion of physiological menstrual cycle levels of estradiol, progesterone, estradiol and progesterone versus the vehicle has been reported. Each hormone was tested in random order, separated by a 1-week period between administrations. Results in the 22 women with complete cross-over data showed that forearm blood flow reliably increased on all interventions during higher acetylcholine doses, but that progesterone induced significantly greater flow than vehicle control (P<0.01). Forearm blood flow on estradiol alone or estradiol combined with progesterone increased blood flow; however, this increase was not significantly greater compared to vehicle control.
Endothelial function was assessed during a double-blind short-term RCT of oral micronized progesterone in early menopausal women (1–11 years since final menstruation) with problematic hot flushes/flashes and night sweats (vasomotor symptoms, VMS). Although there were baseline VMS associations with cardiovascular markers, there were no prospective ones. Participating women were screened to be without historical, clinical or laboratory evidence of cardiovascular disease. During a 3-month RCT of oral micronized progesterone (300 mg at bedtime daily to ensure the blood progesterone concentrations remain above the luteal phase threshold for 24 hours), endothelial function was assessed in 34 women before and after 3 months on experimental therapy. At baseline the 18 women on progesterone did not differ from the 16 women on placebo. Although the forearm blood flow increases on progesterone were consistently greater than on placebo (Figure 4), and they were similar (about 15% greater than placebo) to results of a previous intra-arterial progesterone study, they did not reach statistical significance versus placebo.
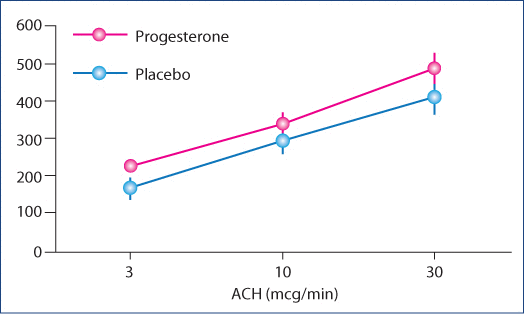
Figure 4:This diagram illustrates the percentage increase (±SE) in forearm blood flow (Y axis) (adjusted for the opposite arm) by venous occlusion plethysmography on active progesterone (300 mg/day for 3 months) or placebo during increasing doses of acetylcholine (ACH). Reprinted with permission of PLOS One from Prior et al.
It is important to consider whether or not stress hormones directly adversely influence endothelial function as the mechanism for ovulatory disturbances appears to be that stressors perceived in the hypothalamus upregulate corticotrophin releasing hormone to both increase pituitary ACTH and adrenal cortisol production. Acting through stressors (higher corticosteroid levels or the catecholamines, epinephrine and norepinephrine) and complex neuroendocrine changes resulting from disturbances in sleep or circadian rhythms there are a series of coordinated actions leading to the ovulatory disturbances. However, it may be that direct effects of cortisol or catecholamines have negative cardiovascular effects. In a randomized double-blind cross-over trial in eight men given high dose glucocorticoid, cortisol (20 mg every 6 hours) for 5 days, venous occlusion plethysmography showed that cortisol completely eliminated the acetylcholine-related increased forearm blood flow. This suggests that adrenal stress hormones have direct negative effects on endothelial function and thus the cardiovascular system; high cortisol may potentiate the adverse cardiovascular effects of insufficient progesterone. In order to confirm this potential endothelial effect interaction, a study of high dose cortisol treatment with and without progesterone in women is required.
EFFECT ON PROGESTERONE BIOMARKERS OF CARDIOVASCULAR RISK:
In the progesterone versus placebo RCT described above, smokers, those with diabetes, hypertension, previous cardiac disease, abnormal cardiograms or abnormal fasting glucose or lipids were excluded. Using the Framingham General Cardiovascular Risk Profile for women, at baseline mean 10-year risks for AMI, heart failure, stroke or peripheral vascular disease were low at about 4%. Results of this study showed that progesterone caused no negative within-woman changes adjusting for placebo therapy (by analysis of covariance) in weight, body mass index, systolic or diastolic blood pressure, heart rate, waist circumference, fasting glucose, total cholesterol, low density lipoprotein cholesterol, triglycerides, C-reactive protein, serum albumen or D-dimer. The small (7.7%) decrease in high density lipoprotein cholesterol (although used to calculate the Framingham General Cardiovascular Risk Profile) did not result in any changes in this metric during the trial, nor in differences in change between progesterone and placebo.
THE CARDIOVASCULAR ELECTRICAL CONDUCTION SYSTEM:
Atrial fibrillation is an abnormal heart rhythm that is rare in those under the age of 55 but common in older individuals. It leads to congestive heart failure, strokes (without effective anticoagulation) and is a significant part of women’s CVD although atrial fibrillation is less common in women than in men. Atrial fibrillation developed in about 1200 of the approximately 27,000 women participating in the two hormone trials of the Women’s Health Initiative. Atrial fibrillation developed more frequently in the hormone-treated versus the placebo-treated women overall, but in the estrogen-only treated women the risk ratio for this incident arrhythmia was significantly increased to 1.17 (95% CI 1.00, 1.36). However, in the estrogen-progestin treated women the incidence of new atrial fibrillation was 1.07 (95% CI 0.91, 1.25) P=0.44.
It is also likely that ovarian hormonal changes are directly responsible for the higher risk for arrhythmias in perimenopausal women. This is a normal life transition when estradiol levels are higher and progesterone levels are lower than in premenopause. Given the experimental data showing that estradiol lengthens the QTc and progesterone shortens it by improving cardiac repolarization, this may be an important CVS mechanism through which progesterone counterbalances the actions of estradiol.
MULTIPLE SPONTANEOUS EARLY MISCARRIAGES:
Spontaneous miscarriages typically occur between 10 and 15 weeks of gestation around which time in the course of pregnancy progesterone production changes from the corpus luteum to the placenta. A large prospective cohort study documented psychosocial, nutritional and hormonal events in over one thousand urban community women in early gestation. Sufficient data were available to analyze the contributing factors to early miscarriage in 55 women with spontaneous pregnancy loss compared with 809 controls delivering at term. Results showed that a lower body weight (body mass index <20 kg/m) and lower serum progesterone levels (<38 nmol/L) as well as being over age 33 were associated with these early miscarriages. Furthermore, these women miscarrying early in pregnancy reported, prior to that event, perceived higher life stresses and demands (and had higher corticotrophin releasing hormone levels). Thus early miscarriages are related to lower progesterone and higher stress hormone levels. Both lower progesterone and higher stress hormone levels are associated with vascular abnormalities as previously discussed.
In a cross-sectional study investigators assessed miscarriages by questionnaire in a representative sample of almost 4000 Finnish woman aged 20–90. Among parous women aged 50–74, having experienced a miscarriage, after age-adjustment, there was an odds ratio of 2.1 (95% CI 1.0–4.3) for clinical cardiovascular disease. The strongest data are prospective and are provided by a large population-based study centred in Heidelberg, Germany and part of the European Prospective Investigation into Cancer and Nutrition (EPIC). This study followed almost 12,000 women who had ever been pregnant for a mean period of 10.8 years; 82 myocardial infarctions were documented during this time. Women with more than three miscarriages had a multiply-adjusted five-fold increased risk for acute myocardial infarction (adjusted HR 5.06 [95% CI 1.26–20.29]. Thus these data suggest that women with lower progesterone levels, lower BMI levels, higher premenopausal age and higher stressors are at increased risk for myocardial infarctions. These data support the hypothesis that normal progesterone levels (as well as normal estradiol levels) are needed for cardiovascular protection in women.
ANOVULATION IN A POPULATION-BASED PROSPECTIVE STUDY:
As part of a breast screening programme in Utrecht, Netherlands a nested case-referent population-based study of risks for early myocardial infarction was performed in women aged 40–49 who continued to menstruate (were non-menopausal and not on hormones). This study enrolled 10,583 women (participation rate 44%) and observed the rate of incident AMI over the subsequent 5–7 years. All women were asked to provide first morning urine samples on cycle day 22 for three consecutive menstrual cycles and to keep a menstrual calendar. All 10 regional hospitals’ records were searched prospectively and 45 women were found to have been admitted for an AMI or chest pain with documented angiographic coronary artery disease. Each of these women with CVD (cases) was matched with three similar-aged women (controls) who had collected urine samples within 6 months of them. Urine samples for cases and referents were analyzed for excretion of estrone, PdG and testosterone and were adjusted for urinary creatinine. Age at menopause was similar for controls and cases. Contrary to the investigators’ expectations, lower estrogen or higher testosterone excretions were not present; urinary PdG levels were also not different. When a criterion for ovulation (PdG ≥0.5 μmol/mmol creatinine) was applied to the dataset to identify participants with any specimens from anovulatory cycles, significantly more women who suffered an MI (29%) than controls (14%) were identified as anovulatory (P<0.02). This evidence supports a role for anovulation during ages 40–49 within regular cycles as important in the risk for early heart attacks in menopausal women.
OVERVIEW
This review suggests that progesterone has beneficial cardiovascular physiological effects and its presence within ovulatory menstrual cycles is associated with decreased risks for women’s cardiovascular diseases. In most instances, clinicians are currently unaware of whether or not normal progesterone levels are present within clinically normal menstrual cycles and lack appreciation that “subclinical” or “silent” ovulatory disturbances are very prevalent. Normal menstrual cycle estradiol levels are clearly important for women’s heart health. However, emerging data on the prevalence and incidence of silent ovulatory disturbances within regular cycles and new RCT data on progesterone’s positive endothelial actions with a lack of adverse effects on surrogate markers for cardiovascular disease have prompted this new hypothesis related to risks for cardiovascular disease in women in menopause.
The reviewed data from basic science, animal experiments, clinical, epidemiological and RCTs all point toward a positive effect of ovulation and the high post-ovulatory levels of progesterone (in the context of normal estradiol levels) on various aspects of women’s cardiovascular health. In some circumstances the effects of estradiol may be negative (as in lengthening the QTc interval) and evidence suggests that progesterone counterbalances this. In endothelial function, estradiol and progesterone appear to both indirectly increase blood flow through the nitric oxide system. Progesterone also has direct vasodilatory actions through vascular smooth muscle.
This review is strengthened by the breadth of the information assessed – from in vitro and basic science, to animal studies to clinical experimental and epidemiological data. Furthermore, this review may be beneficial in terms of integration of information into clinically sensible and physiologically plausible relationships. However, there are limitations to this review that warrant mention including that the review is by no means comprehensive but merely highlights possible relationships. Any omissions are not by design but by lack of awareness or inability to make integrated sense of the data.
Although it is early in the development of these ideas, there are both research and clinical options to explore and validate them. The research implications of this hypothesis are: 1) there is a need for a non-invasive but accurate test of ovulation that does not require cycle-phase timing; 2) women identified as having ovulatory disturbances (as in with non-male factor or obstructive infertility) or low progesterone levels (as in recurrent miscarriages) could to be prospectively observed, along with suitable control groups, for the development of arrhythmias and other cardiovascular diseases; 3) women with proven frequent ovulatory disturbances could be randomized to cyclic progesterone therapy or a placebo and observed for development of changes in cardiovascular markers.
Likewise, there are many clinical implications of this new concept about risk factors for women’s cardiovascular disease including: 1) in premenopausal women experiencing hypothalamic amenorrhea, oligomenorrhea or irregular cycles, in addition to dealing with each woman’s social, environmental and nutritional stressors, it is worth considering cyclic progesterone therapy (http://www.cemcor.ubc.ca/help_yourself/articles/cyclic_progesterone_therapy) to prevent bone loss and potentially cardiovascular disease; 2) women identified as having had more than one early miscarriage could be “prophylactically” treated with progesterone for the first 12 weeks of a confirmed pregnancy; and 3) women identified as having ovulatory disturbances causing infertility be treated with cyclic progesterone (begun after the LH peak); 4) perimenopausal women presenting to emergency rooms or physicians with inexplicable chest pain or arrhythmias could be treated with cyclic or daily progesterone.
CONCLUSION
Menopause does not suddenly increase women’s risks for heart attacks. Estrogen treatment, even as a transdermal application (having fewer thrombotic risks than oral), and when provided early in menopause (if the higher risks of breast cancer are ignored), is not a logical solution to prevent women’s heart disease. Prevention of women’s cardiovascular disease begins during the premenopausal and perimenopausal years and requires not just estradiol but also progesterone. Ovulatory disturbances (as only briefly touched on here) arise because of social inequities, abuse, inadequate caloric intake to balance energy needs, illness, over-exercise and other “threats” or stressors. Thus, for women to be normally ovulatory they need to feel “at home” in their social, emotional, physical, nutritional and spiritual environments.
The hypothesis that normally ovulatory cycles will prevent women’s cardiovascular disease integrates socio-cultural and stress-related variables with traditional cardiovascular risk factors. Further, this new hypothesis demands an innovative, multifaceted women’s CHD prevention strategy. To be implemented it would require elimination of gender-based economic and social inequities and stereotypes and to combine social support, cognitive behavioural therapy, optimization of nutrition and activity with management of traditional obesity, insulin resistance/ diabetes, cigarette addiction and inactivity risk factors. In addition, the availability of oral progesterone as a pharmaceutical product, such as oral micronized progesterone, provides a possible cyclic therapy if these other strategies are unsuccessful (for example, in perimenopause). Oral progesterone also has advantageous “side effects” of improving sleep, effectively treating hot flushes/flashes and night sweats and possibly decreasing anxiety. Therefore, if anovulation or short luteal phase cycles are discovered, and premenstrual progesterone levels are low, then it is possible to provide “luteal phase replacement therapy” by treatment with cyclic oral micronized progesterone and to know that the estradiol-progesterone imbalance that is likely related to cardiovascular risks, as well as to negative bone mineral density changes may be resolved.
DISCLOSURE OF INTERESTS
REFERENCES
- Tunstall-Pedoe H. Myth and paradox of coronary risk and the menopause.
- Anand SS, Islam S, Rosengren A, Franzosi MG, Steyn K, Yusufali AH, et al. Risk factors for myocardial infarction in women and men: insights from the INTERHEART study.
- Pilote L, Dasgupta K, Guru V, Humphries KH, McGrath J, Norris C, et al. A comprehensive view of sex-specific issues related to cardiovascular disease.
- Mosca L, Banka CL, Benjamin EJ, Berra K, Bushnell C, Dolor RJ, et al. Evidence-based guidelines for cardiovascular disease prevention in women: 2007 update.
- Heron M. Leading Causes of Death 2009.
- Izadnegahdar M, Singer J, Lee MK, Gao M, Thompson CR, Kopec J, et al. Do younger women fare worse? Sex differences in acute myocardial infarction hospitalization and early mortality rates over ten years.
- Taylor F, et al. Statins for the primary prevention of cardiovascular disease.
- Ridker PM, Cook NR, Lee IM, Gordon D, Gaziano JM, Manson JE, et al. A randomized trial of low-dose aspirin in the primary prevention of cardiovascular disease in women.
- Kannel WB, McGee DL. Diabetes and cardiovascular disease. The Framingham study.
- Levitzky YS, Pencina MJ, D’Agostino RB, Meigs JB, Murabito JM, Vasan RS, et al. Impact of impaired fasting glucose on cardiovascular disease: the Framingham Heart Study.
- Stampfer MJ, Golditz GA, Willett WC, Manson JE, Rosner B, Speizer FE, et al. Postmenopausal estrogen therapy and cardiovascular disease.
- Baxter S, Prior JC.
- Prior JC. Postmenopausal estrogen therapy and cardiovascular disease (letter).
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in health postmenopausal women: principal results from the Women’s Health Initiative Randomized Control trial.
- Anderson GL, Limacher M, Assaf AR, Bassford T, Beresford SA, Black H, et al. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial.
- Harman SM, Brinton EA, Cedars M, Lobo R, Manson JE, Merriam GR, et al. KEEPS: the kronos early estrogen prevention study.
- Barrett-Connor E. Sex differences in coronary heart disease. Why are women so superior? The 1995 Ancel Keys Lecture.
- Li D, Hitchcock CL, Barr SI, Yu T, Prior JC. Negative spinal bone mineral density changes and subclinical ovulatory disturbances–prospective data in healthy premenopausal women with regular menstrual cycles.
- Prior JC, Elliott TG, Norman E, Stajic V, Hitchock CL. Progesterone therapy, endothelial function and cardiovascular risk factors: a 3-month randomized, placebo-controlled trial in healthy early postmenopausal women.
- Prior JC, Naess M, Langhammer A, Forsmo S. The point prevalence of ovulation in a large population-based sample of spontaneously, regularly menstruating women. The HUNT Study. Norway,
- Vollman RF. The menstrual cycle. In: Friedman EA, ed.
- Vitzthum VJ. The ecology and evolutionary endocrinology of reproduction in the human female.
- Hammar N, Alfredsson L, Rosen M, Spetz CL, Kahan T, Ysberg AS. A national record linkage to study acute myocardial infarction incidence and case fatality in Sweden.
- O’Donnell E, Goodman JM, Harvey PJ. Clinical review: Cardiovascular consequences of ovarian disruption: a focus on functional hypothalamic amenorrhea in physically active women.
- Creinin MD, Keverline S, Meyn LA. How regular is regular? An analysis of menstrual cycle regularity.
- Abraham GE. The normal menstrual cycle. In: Givens JR, ed.
- Hulley S, Bush T, Furberg C, Herrington D, Riggs B, Vittinghoff E. Randomized trial of estrogen plus progestin for secondary prevention of coronary heart disease in postmenopausal women.
- Coronary Drug Project Research Group. Coronary drug project: initial findings leading to modifications of its research protocol.
- Coronary Drug Project Research Group. Coronary drug project: findings leading to the discontinuation of the 2.5 mg/day estrogen group.
- Malcolm CE, Cumming DC. Does anovulation exist in eumenorrheic women?
- MacDonald PC, Dombroski RA, Casey ML. Recurrent secretion of progesterone in large amounts: an endocrine/metabolic disorder unique to young women?
- Vitzthum VJ, Thornburg J, Spielvogel H. Seasonal modulation of reproductive effort during early pregnancy in humans.
- Prior JC, Vigna YM, Schechter MT, Burgess AE. Spinal bone loss and ovulatory disturbances.
- Sowers M, Randolph JF, Crutchfield M, Jannausch ML, Shapiro B, Zhang B, et al. Urinary ovarian and gonadotropin hormone levels in premenopausal women with low bone mass.
- Stachenfeld NS, Silva C, Keefe DL. Estrogen modifies the temperature effects of progesterone.
- Israel R, Mishell DR, Jr., Stone SC, Thorneycroft IH, Moyer DL. Single luteal phase serum progesterone assay as an indicator of ovulation.
- Vuorento T, Hovatta O, Kurunmaki H, Ratsula K, Huhtaniemi I. Measurements of salivary progesterone throughout the menstrual cycle in women suffering from unexplained infertility reveal high frequency of luteal phase defects.
- Kassam A, Overstreet JW, Snow-Harter C, De Souza MJ, Gold EB, Lasley BL. Identification of anovulation and transient luteal function using a urinary pregnanediol-3-glucuronide ratio algorithm.
- O’Connor KA, Brindle E, Miller RC, Shofer JB, Ferrell RJ, Klein NA, et al. Ovulation detection methods for urinary hormones: precision, daily and intermittent sampling and a combined hierarchical method.
- Prior JC, Vigna YM, Schulzer M, Hall JE, Bonen A. Determination of luteal phase length by quantitative basal temperature methods: validation against the midcycle LH peak.
- Bedford JL, Prior JC, Hitchcock CL, Barr SI. Detecting evidence of luteal activity by least-squares quantitative basal temperature analysis against urinary progesterone metabolites and the effect of wake-time variability.
- Shepard MK, Senturia YD. Comparison of serum progesterone and endometrial biopsy for confirmation of ovulation and evaluation of luteal function.
- Landgren BH, Unden AL, Diczfalusy E. Hormonal profile of the cycle in 68 normally menstruating women.
- Di Carlo C, Shaham Z, MacDougall J, Patel A, Hall ML, Jacobs HS. Polcystic ovaries as a protective factor for bone mineral loss in young women with amenorrhea.
- Hahn S, Janssen OE, Tan S, Pleger K, Mann K, Schedlowski M, et al. Clinical and psychological correlates of quality-of-life in polycystic ovary syndrome.
- Bedford JL, Prior JC, Barr SI. A prospective exploration of cognitive dietary restraint, subclinical ovulatory disturbances, cortisol and change in bone density over two years in healthy young women.
- Nielsen HK, Brixen K, Bouillon R, Mosekilde L. Changes in biochemical markers of osteoblastic activity during the menstrual cycle.
- Clarke CL, Sutherland RL. Progestin regulation of cellular proliferation.
- Groshong SD, Owen GI, Grimison B, Schauer IE, Todd MC, Langan TA, et al. Biphasic regulation of breast cancer cell growth by progesterone: role of the cyclin-dependent kinase inhibitors, p21 and p27(Kip1).
- Zhang X, Jeyakumar M, Bagchi MK. Ligand-dependent cross-talk between steroid and thyroid hormone receptors. Evidence for common transcriptional coactivator(s).
- Graham JD, Clarke CL. Physiological action of progesterone in target tissue.
- Rickard DJ, Waters KM, Ruesink TJ, Khosla S, Katzenellenbogen JA, Katzenellenbogen BS, et al. Estrogen receptor isoform-specific induction of progesterone receptors in human osteoblasts.
- Hitchcock CL, Prior JC. Oral Micronized Progesterone for Vasomotor Symptoms in Healthy Postmenopausal Women – a placebo-controlled randomized trial.
- Ishida Y, Killinger DW, Khalil MW, Yang K, Strutt B, Heersche JN. Expression of steroid-converting enzymes in osteoblasts derived from rat vertebrae.
- Ishida Y, Heersche JN. Progesterone stimulates proliferation and differentiation of osteoprogenitor cells in bone cell populations derived from adult female but not from adult male rats.
- Dubey RK, Gillespie DG, Jackson EK, Keller PJ. 17Beta-estradiol, its metabolites, and progesterone inhibit cardiac fibroblast growth.
- Morey AK, Pedram A, Razandi M, Prins BA, Hu RM, Biesiada E, et al. Estrogen and progesterone inhibit vascular smooth muscle proliferation.
- Everaert BR, Van Craenenbroeck EM, Hoymans VY, Haine SE, Van NL, Conraads VM, et al. Current perspective of pathophysiological and interventional effects on endothelial progenitor cell biology: focus on PI3K/AKT/eNOS pathway.
- Lemieux C, Cloutier I, Tanguay JF. Menstrual cycle influences endothelial progenitor cell regulation: a link to gender differences in vascular protection?
- Hoetzer GL, MacEneaney OJ, Irmiger HM, Keith R, Van Guilder GP, Stauffer BL, et al. Gender differences in circulating endothelial progenitor cell colony-forming capacity and migratory activity in middle-aged adults.
- Tanaka S, Ueno T, Sato F, Chigusa Y, Kawaguchi-Sakita N, Kawashima M, et al. Alterations of circulating endothelial cell and endothelial progenitor cell counts around the ovulation.
- Simoncini T, Mannella P, Fornari L, Caruso A, Willis MY, Garibaldi S, et al. Differential signal transduction of progesterone and medroxyprogesterone acetate in human endothelial cells.
- Barbagallo M, Dominguez LJ, Licata G, Shan J, Bing L, Karpinski E, et al. Vascular effects of progesterone: role of cellular calcium regulation.
- Moldovanova I, Schroeder C, Jacob G, Hiemke C, Diedrich A, Luft FC, et al. Hormonal influences on cardiovascular norepinephrine transporter responses in healthy women.
- Odening KE, Choi BR, Liu GX, Hartmann K, Ziv O, Chaves L, et al. Estradiol promotes sudden cardiac death in transgenic long QT type 2 rabbits while progesterone is protective.
- Landau RL, Bergenstal DM, Lugibihl K, Kascht ME. The metabolic effects of progesterone in man.
- Barr SI, Janelle KC, Prior JC. Energy intakes are higher during the luteal-phase of ovulatory menstrual cycles.
- Frisch RE, McArthur JW. Menstrual cycles: fatness as a determinant of minimum weight for height necessary for their maintenance or onset.
- Jasienska G, Ellison PT. Energetic factors and seasonal changes in ovarian function in women from rural Poland.
- Yeung EH, Zhang C, Mumford SL, Ye A, Trevisan M, Chen L, et al. Longitudinal study of insulin resistance and sex hormones over the menstrual cycle: the BioCycle Study.
- Bagis T, Gokcel A, Zeyneloglu HB, Tarim E, Kilicdag EB, Haydardedeoglu B. The effects of short-term medroxyprogesterone acetate and micronized progesterone on glucose metabolism and lipid profiles in patients with polycystic ovary syndrome: a prospective randomized study.
- Shaikh AA, Naqvi RH, Shaikh SA. Concentrations of oestradiol-17-beta and progesterone in peripheral plasma of cynomolgus monkey (Macaca fascicularis) in relation to length of menstrual cycle and its component phases.
- Kaplan JR, Adams MR, Clarkson TB, Koritnik DR. Psychological influences on female ‘protection’ among cynomolgus macaques.
- Adams MR, Kaplan JF, Koritnik DR. Ovariectomy, social status, and atherosclerosis in cynomolgus monkeys.
- Kaplan JR, Adams MR, Koritnik DR, Rose JC, Manuck SB. Adrenal responsiveness and social status in intact and ovariectomized Macaca fascicularis.
- Minshall RD, Stanczyk FZ, Miyagawa K, Uchida B, Axthelm M, Novy M, et al. Ovarian steroid protection against coronary artery hyperreactivity in rhesus monkeys.
- Mangos GJ, Walker BR, Kelly JJ, Lawson JA, Webb DJ, Whitworth JA. Cortisol inhibits cholinergic vasodilation in the human forearm.
- Prior JC, Ho Yeun B, Clement P, Bowie L, Thomas J. Reversible luteal phase changes and infertility associated with marathon training.
- Selye H. The effect of adaptation to various damaging agents on the female sex organs in the rat.
- Prior JC. Endocrine “conditioning” with endurance training: a preliminary review.
- Prior JC. Exercise-associated menstrual disturbances. In: Adashi EY, Rock JA, Rosenwaks Z, eds.
- Barr SI, Janelle KC, Prior JC. Vegetarian versus nonvegetarian diets, dietary restraint, and subclinical ovulatory disturbances: prospective six month study.
- McLean JA, Barr SI, Prior JC. Cognitive dietary restraint is associated with higher urinary cortisol excretion in healthy premenopausal women.
- Prior JC, Seifert-Klauss VR, Hale G. The endocrinology of perimenopause – new definitions and understandings of hormonal and bone changes. In: Dvornyk V, ed.
- Prior JC. The ageing female reproductive axis II: ovulatory changes with perimenopause. In: Chadwick DJ, Goode JA, eds.
- Weiss G, Skurnick JH, Goldsmith LT, Santoro NF, Park SJ. Menopause and hypothalamic-pituitary sensitivity to estrogen.
- La Vecchia C, Decarli A, Franceschi S, Gentile A, Negri E, Parazzini F. Menstrual and reproductive factors and the risk of myocardial infarction in women under fifty-five years of age.
- Palmer JR, Rosenberg L, Shapiro S. Reproductive factors and risk of myocardial infarction.
- Bertuccio P, Tavani A, Gallus S, Negri E, La Vecchia C. Menstrual and reproductive factors and risk of non-fatal acute myocardial infarction in Italy.
- Harris MA, Prior JC, Koehoorn M. Age at menarche in the Canadian population: secular trends and relationship to adulthood BMI.
- Jacobsen BK, Heuch I, Kvale G. Association of low age at menarche with increased all-cause mortality: a 37-year follow-up of 61,319 Norwegian women.
- Jacobsen BK, Nilssen S, Heuch I, Kvale G. Does age at natural menopause affect mortality from ischemic heart disease?
- Kalyan S, Hitchcock CL, Sirrs S, Pudek M, Prior JC. Cardiovascular and metabolic effects of medroxyprogesterone acetate versus conjugated equine estrogen after premenopausal hysterectomy with bilateral ovariectomy.
- Solomon CG, Hu FB, Dunaif A, Rich-Edwards JE, Stampfer MJ, Willett WC, et al. Menstrual cycle irregularity and risk for future cardiovascular disease.
- Talbott EO, Kuller LH, Detre K, Matthews K, Norman S, Kelsey SF, et al. Reproductive history of women dying of sudden cardiac death: a case-controlled study.
- Lawlor DA, Emberson JR, Ebrahim S, Whincup PH, Wannamethee SG, Walker M, et al. Is the association between parity and coronary heart disease due to biological effects of pregnancy or adverse lifestyle risk factors associated with child-rearing? Findings from the British Women’s Heart and Health Study and the British Regional Heart Study.
- Catov JM, Newman AB, Sutton-Tyrrell K, Harris TB, Tylavsky F, Visser M, et al. Parity and cardiovascular disease risk among older women: how do pregnancy complications mediate the association?
- Parikh NI, Cnattingius S, Dickman PW, Mittleman MA, Ludvigsson JF, Ingelsson E. Parity and risk of later-life maternal cardiovascular disease.
- Hitchcock CL, Elliott TG, Norman EG, Stajic V, Teede H, Prior JC. Hot flushes and night sweats differ in associations with cardiovascular markers in healthy early postmenopausal women.
- Mather KJ, Norman EG, Prior JC, Elliott TG. Preserved forearm endothelial responses with acute exposure to progesterone: a randomized cross-over trial of 17-b estradiol, progesterone, and 17-b estradiol with progesterone in healthy menopausal women.
- Simon JA, Shangold MM, Andrews MC, Buster JC, Hodgen GD. Micronized progesterone therapy: the importance of route of administration and pharmacokinetics on clinical outcome.
- Berga SL, Daniels TL, Giles DE. Women with functional hypothalamic amenorrhea but not other forms of anovulation display amplified cortisol concentrations.
- Williams WP, III, Kriegsfeld LJ. Circadian control of neuroendocrine circuits regulating female reproductive function.
- D’Agostino RB, Sr., Vasan RS, Pencina MJ, Wolf PA, Cobain M, Massaro JM, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study.
- Perez MV, Wang PJ, Larson JC, Virnig BA, Cochrane B, Curb JD, et al. Effects of postmenopausal hormone therapy on incident atrial fibrillation: the Women’s Health Initiative randomized controlled trials.
- Santoro N, Rosenberg J, Adel T, Skurnick JH. Characterization of reproductive hormonal dynamics in the perimenopause.
- Prior JC. Perimenopause: the complex endocrinology of the menopausal transition.
- Arck P, Hansen PJ, Mulac JB, Piccinni MP, Szekeres-Bartho J. Progesterone during pregnancy: endocrine-immune cross talk in mammalian species and the role of stress.
- Arck PC, Rucke M, Rose M, Szekeres-Bartho J, Douglas AJ, Pritsch M, et al. Early risk factors for miscarriage: a prospective cohort study in pregnant women.
- Kharazmi E, Fallah M, Luoto R. Miscarriage and risk of cardiovascular disease.
- Kharazmi E, Dossus L, Rohrmann S, Kaaks R. Pregnancy loss and risk of cardiovascular disease: a prospective population-based cohort study (EPIC-Heidelberg).
- Gorgels WJ, Graaf Y, Blankenstein MA, Collette HJ, Erkelens DW, Banga JD. Urinary sex hormone excretions in premenopausal women and coronary heart disease risk: a nested case-referent study in the DOM-cohort.
- Prior JC, Vigna YM, Barr SI, Rexworthy C, Lentle BC. Cyclic medroxyprogesterone treatment increases bone density: a controlled trial in active women with menstrual cycle disturbances.
- Canonico M, Oger E, Plu-Bureau, Conard J, Meyer G, Levesque H, et al. Hormone therapy and venous thromboembolism among postmenopausal women: impact of the route of estrogen administration and progestogens: the ESTHER study.
- Schussler P, Kluge M, Yassouridis A, Dresler M, Held K, Zihl J, et al. Progesterone reduces wakefulness in sleep EEG and has no effect on cognition in healthy postmenopausal women.
- Prior JC, Hitchcock CL. Progesterone for hot flush and night sweat treatment – effectiveness for severe vasomotor symptoms and lack of withdrawal rebound.
- Le Mellédo JM, Baker G. Role of progesterone and other neuroactive steroids in anxiety disorders.
- Dennerstein L, Spencer-Gardner C, Gotts G, Brown JB, Smith MA. Progesterone and the premenstrual syndrome: a double blind crossover trial.
- Waugh EJ, Polivy J, Ridout R, Hawker GA. A prospective investigation of the relations among cognitive dietary restraint, subclinical ovulatory disturbances, physical activity, and bone mass in healthy young women.
- Morris FL, Payne WR, Wark JD. The impact of intense training on endogenous estrogen and progesterone concentrations and bone mineral acquisition in adolescent rowers.
- Waller K, Reim J, Fensler L, Swan SH, Brumback B, Windham GC, et al. Bone mass and subtle abnormalities in ovulatory function in healthy women.
- Prior JC, Vigna YM, Barr SI, Kennedy S, Schulzer M, Li DK. Ovulatory premenopausal women lose cancellous spinal bone: a five year prospective study.