Abstract
Estriol, coined the “weaker” of the three endogenous estrogens, has significant therapeutic effects, some of which are little known to clinicians. Estriol provides numerous clinical benefits, commanding the attention of researchers dating as far back as 1966 and continues to garner substantial consideration as a valuable and viable therapeutic agent. Some of the most common and effective treatments that employ estriol include: hot flashes, insomnia, skin enhancement, vaginal atrophy and reduced frequency of urinary tract infections. Most recently, estriol has shown the potential to treat individuals with Th1-mediated autoimmune illnesses, including multiple sclerosis and rheumatoid arthritis. This review article will update the clinical effects and benefits of estriol and further clarify the documented advances which support the substantial therapeutic benefits of estriol for autoimmune conditions. The availability of compounded estriol preparations will also be addressed.
INTRODUCTION
Due to the well-known and documented risks associated with estradiol (E2), estriol (E3) has long piqued the interest of researchers and clinicians. For decades, E3 has proven its efficacy in the successful management of menopausal symptoms. What none of these early pioneers could have predicted however, was that treatment with E3 would provide breakthroughs as an agent in the treatment of Th1-mediated autoimmune disease. E3, for its protective and clinically beneficial actions, is considered a cornerstone in the repertoire of bio-identical hormone therapies (BHT). E2, the well-known, well-studied, and most prescribed form of estrogen, is usually referred to synonymously with estrogen, due to its stronger actions and ubiquitous familiarity as the common estrogen-replacement therapy (ERT). However, E3 is unique as an “estrogen” due to its antagonistic effects on E2 in breast tissue. Dr. H. M. Lemon, University of Nebraska Medical Center in Omaha, noted throughout his extensive investigative career that women who developed breast cancer had significantly lower levels of E3 than women who did not develop breast cancer. Lemon also reviewed six studies which found that the estrogen quotient (EQ) was higher in populations with a lower risk of breast cancer. Lemon further elucidated that E3 had a place in both the treatment of menopausal women and for the prevention of mammary carcinoma. These findings and others have become even more important and relevant following the release of the Women’s Health Initiative (WHI) in 2002, which revealed significant and dangerous health risks associated with hormone replacement therapy (HRT). This raised concerns with conventional ERT and HRT, and resulted in a dramatically raised consciousness and increased awareness in patients with breast cancer and for women in general, and further emphasized the need for hormone therapies with little or fewer risks.
BACKGROUND
PHYSIOLOGY OF ESTROGENS:
Estrogens are steroid hormones present in both men and women, but present at significantly higher levels in women of reproductive age. There are three naturally occurring estrogens in women: estrone (E1), E2, and E3. The developing follicles in the ovaries are responsible for producing the majority of the estrogens. There is also evidence that some estrogens are formed by the liver, adrenal glands, muscle tissue and fat cells through conversion of C19 precursors to C18 steroids by aromatase. Adipose tissue expression of aromatase increases with body weight and increasing age, thus is of concern in in post-menopausal women. Synthesis of estrogens occurs in the theca interna cells of the developing ova, and is accomplished through conversion of androstenedione from cholesterol. E2 can be converted to E1 and E3. E1 can be converted to E2 or E3. However, E3 is not interconvertible and hence, does not result in an increase of either of the other two estrogens (Figure 1).
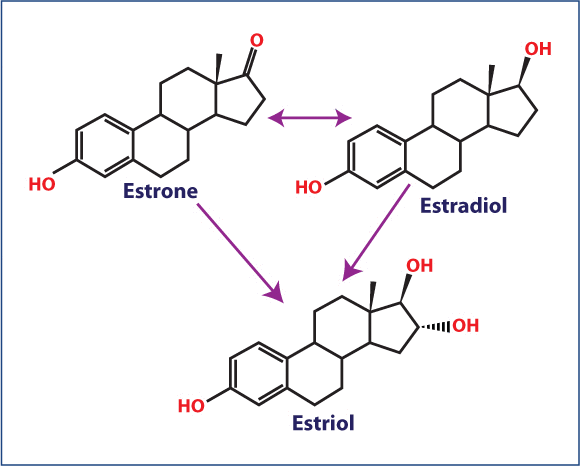
Interconversion of the three naturally occurring estrogens. Estrone (E1) and estradiol (E2) can be converted to all three forms, but estriol (E3) is not interconvertible
ESTROGEN QUOTIENT
Lemon observed a relationship between the protective effects of E3 and the relative presence of the other two estrogens. He developed a mathematical formula, which he termed the estrogen quotient (EQ). The EQ is a ratio designed to assess risk for reproductive tissue that would be vulnerable to the proliferative influences of E2. The quotient formula is: E3/(E1 + E2). Lemon arrived at this via his understanding that the sum of the potentially riskier estrogens E1 and E2 (proliferative or carcinogenic estrogens) could be modified and/or overridden by the presence of “enough” E3. Although not a validated measure, the EQ may provide a laboratory measure to predict a woman’s risk and aid in monitoring the success of a patient’s therapies designed to alter her estrogen status. The optimal ratio for the breast tissue protection has been established as an EQ >1.5. International studies also document the ratio of E3 to the other estrogens in Asian women, and compares this quotient to North American women both in follicular and luteal phases. The findings demonstrate considerably higher EQs in Asian women, consistent with the lower rates of breast cancer in corresponding areas of Asia.
SUCCESSFUL MANAGEMENT OF MENOPAUSAL SYMPTOMS
E3 has been successfully employed for decades to address the symptoms of menopause, including the safe and effective management of hot flashes, night sweats, insomnia, and genital atrophy. There is a substantial body of literature that explores the concern of endometrial hyperplasia, the unwanted growth of the uterus lining, which is a known risk of E2 supplementation with E3 use. In all of the studies reviewed, the use of E3 as a therapeutic agent did not increase the risk of endometrial or ovarian cancers, nor did it cause endometrial hyperplasia. Of additional note, E3 has been reported to lower blood pressure and improve lipid metabolism. A further application of E3 that needs more exploration is for the treatment of uterine prolapse due to loss of pelvic floor integrity. One study revealed promising beneficial results when E3 was added to the rehabilitation protocol.
AUTOIMMUNITY AND IMMUNE MODULATION
Endogenous E3 levels during pregnancy being up to four times higher than in non-pregnant women has a positive impact on modulating immunity and improving symptoms of some autoimmune conditions, including multiple sclerosis (MS) and rheumatoid arthritis (RA). Sex hormones have been linked to differences in prevalence of autoimmune diseases such as MS between genders. MS is more prevalent in women than in men, possibly due to a protective effect offered by testosterone. In women, during late pregnancy, there is a decrease in disease severity when estriol levels are highest, suggesting that estriol may offer some degree of protection in women.
Although clinical evidence in patients suffering from autoimmune diseases is lacking, several in vitro and in vivo studies have investigated the relationship between estrogens and auto-immune disease. Jansson et al. reported the modulation of the course of chronic, relapsing encephalomyelitis (EAE) in a mouse model by E2 and E3. Castrated female mice were challenged with myelin basic protein (MBP) and developed EAE, a condition very similar to MS in humans. Treatment of MBP-challenged mice with either E2 or E3 displayed a significant delay in the onset of EAE. Also noted in this study was the major advantage of the utilization of E3 over E2 due to an additional delaying of disease onset by 1 week versus 4 weeks. Furthermore, the investigators noted the suppression of collagen-induced arthritis (CIA) by E2 and E3 treatments. These findings reveal potential promise for ameliorating Th1-cell-dependent autoimmune diseases (MS and RA) with these estrogens.
The pursuit of further validity and scientific understanding of E3’s actions as a beneficial therapy then turned to a more intricate look at how the estrogen acts and behaves. Overstimulation of the Th1 helper cell and/or Th17 population is what ultimately leads to autoimmune diseases such as MS and RA. In 1999, Kim et al. dissected the T-lymphocyte system to evaluate the cause of this over-activation. Utilizing an EAE mouse model, the researchers demonstrated a significant increase in Th2 cytokine interleukin (IL)-10 production in the E3-treated mice compared with the placebo group. Th2 lymphocytes suppressed the immune response and provided a balance between Th1 immune stimulation within the lymphocyte populations. IL-10 is the mediating cytokine produced by the Th2 cells. Taken together, the results of these studies suggest that, due to estriol’s impact on modulating immunity, E3 therapy at concentrations observed during pregnancy, may be a potential treatment modality for Th1-mediated autoimmunity.
A successful attempt to further elucidate the role of E3 and its mechanism of action for immune modulation was achieved by Papenfuss et al. This research team exposed dendritic cells (DCs) to E3 in vitro and infused them into mice prior to the induction of EAE. DCs are myeloid in origin and play a major role in the differentiation of Th1 and Th2 cells and hence have great impact on their host’s immune function. This particular research aimed its focus on the activation of T-cells and found that E3 provided protection against the development of EAE in the mice administered exposed DCs. This finding further expands our understanding of E3 and its benefits to now include an effect on DCs.
Zang et al. evaluated the effect of pregnancy levels of E3 on in vitro T-cell function of peripheral blood monocytes (PBMCs) isolated from seven MS patients. Transmigration and nuclear factor (NF)-κB production are components of the inflammatory cascade and cell destruction associated with autoimmune conditions such as MS. This study suggested that transmigration and NF-κB was inhibited by pregnancy levels of E3. This observation highlighted that E3’s clinical impact on autoimmunity may be accomplished by attenuating the inflammatory cascade and preserving cellular integrity.
Further revelations about E3 and its potential benefits were noted by researchers reported on a study analyzing mouse microglia (CNS immune defense function). It was observed that when activated, these microglia produced nitric oxide (NO) and tumor necrosis factor (TNF)-α. NO and TNF-α can be toxic to myelin-producing oliogodendrocyte cells. EAE and MS are diseases associated with the loss of myelin. These researchers demonstrated that by introducing E3, E2 or progesterone into cell cultures, the production of NO and TNF-α by the microglia was inhibited. The dosage of these hormones mimicked the levels produced during pregnancy. These results suggested that E3 inhibits microglia cell activation which may result in a decrease the severity of multiple sclerosis symptoms.
The first human trial using estriol in non-pregnant women diagnosed with relapsing remitting MS (RRMS) by Sicotte et al. in 2002, was highly encouraging. All of the women who were given 8 mg of E3 daily, had a reduction in the number and volume of gadolinium-enhancing lesions in the brain (a gadolinium-enhanced lesion appears on an MRI scan as a bright spot and is an indication of active inflammation). Enhancing lesions are known to represent active white matter destruction. When the E3 dosing was withdrawn, the enhancing lesions returned to pretreatment activity. PMBCs collected from these subjects were evaluated by intracellular cytokine staining and cytometrically for IL-2, IL-4, IL-5, IL-10, TNF-α and IFN-γ. The stimulated PMBCs displayed significant increases in IL-5 and IL-10 and decreased TNF-α; meaning inflammatory parameters were reduced while stabilization of the immune defenses were enhanced. These changes correlated with reduction in the MRI enhancing lesions in the patients with RRMS. These observations confirmed a shift from Th1 to Th2 immunity in females taking E3 with RRMS, which appeared to be protective in MS patients. These results suggested that providing women with doses of estriol, equivalent to those during pregnancy, may be a novel strategy in multiple sclerosis and may have relevance to other autoimmune diseases. More recently, it has been postulated that men may also respond similarly to E3 therapy, however, to date only studies in animals have investigated effects on males.
The growing body of literature provides evidence of the potential beneficial effects of E3 as a therapeutic agent for autoimmunity. In 2009, Gold and Voskuhl summarized their findings and provided a review of the medical literature related to the treatment of MS. In addition to evidence related to the favorable shifts in the immune response when treated with E3, the authors discuss the in vitro and in vivo neuroprotective benefits of E3 and E2 in the mouse EAE model, in which neuroprotection extends to protection of neurons by decreasing apoptosis, preserving elecrophysiologic function and preventing excitotoxicity, and protecting oligodendrocytes against cytotoxicity. The neuroprotective benefits of these estrogens are also evident in animal models of Parkinson’s disease, cerebellar ataxia, late onset leukodystrophy, stroke, and spinal cord injury. This is most often due to reduced apoptosis. Animal studies have also provided additional evidence of the trophic effects on pyramidal cells in the hippocampus. Ziehn et al. demonstrated prevention of the loss of hippocampal synaptic transmission by E3 in the EAE model E3 treatment protected rodent hippocampal cornu ammonis 1 by maintaining pre- and post-synaptic protein levels and preserving synaptic transmission. These results are of importance as loss of hippocampal synaptic transmission is postulated to be a primary factor in impaired learning and memory loss in MS patients. It is expected that more conclusive evidence will be afforded by ongoing phase II/III trials of estrogens in MS patients.
BONE DENSITY
Whether or not E3 provides adequate protection for bone density health remains controversial. Interestingly, oral E3 is the most preferred prescription for climacteric women in Japan. It has been approved as a treatment for osteoporosis under the Japanese national medical insurance coverage. Kikaet al. compared E3 to conjugated equine estrogens (CEE) for bone loss and lipid metabolism. These results clearly demonstrated the overall benefits of E3 for these purposes. The study revealed equal benefits from E3 and CEE, with both demonstrating a clear effect of increasing bone mass density (BMD). However, E3 produced fewer adverse events of uterine bleeding and, unlike CEE did not elevate triglycerides.
SAFETY
E3 presents itself as a safe and effective therapy which affords many benefits, without the risk of conventional ERT. Although use of E3 may not be as prevalent as conventional ERTs, it is important to note that, to date, no adverse effects of E3 have been reported to the FDA. The authors of a 5-year prospective study analyzing the application of estriol succinate to treat patients with climacteric complaints concluded with the following, “estriol succinate is accordingly a very effective and well tolerated preparation against climacteric complaints, exerting no significant side effects.” This study included 911 patients for a period of 5 years, which translated to 2007 “treatment” years. The incidence of all complications was not increased by estriol succinate, but was in fact, even lower than expected. Endometrial and ovarian cancers were not seen. Two mammary cancers occurred during this period, however, they were unlikely related to treatment. Emboli, myocardial infarction, cerebrovascular and liver-gall bladder complications did not occur during the E3 treatment. Although the overall risks of E3 therapy for MS compares most favorably with the current pharmaceuticals, e.g., IFNβ and Glatiamer, which produce significant systemic side effects, there is still the question of increased infectious disease risk. Robinson and Klein make this point in their review article. They suggested that the suppression of cellular immunity which naturally occurs during pregnancy as a result of E3 immune modulation may put women at greater risk of influenza and malaria. The overall impact or manifestation of this risk to MS patients should surface during the ongoing phase II/III clinical trials and is worthy of further consideration. Longer term studies to follow patients on E3 treatment beyond 5 years is warranted to provide further information on its safety profile as treatment duration may increase with increased longevity.
CONCLUSION
E3 is an alternative option to other HRTs for the effective management of menopausal symptoms and may provide clinical benefits equal to or exceeding that of CEEs or E2 without significant risk. E3 may be an effective immune modulator due to its ability to shift the pathological Th1 responses toward immune down-regulation by the Th2 arm of the immune system. For this reason, E3 warrants consideration for clinical application in the treatment of Th1 autoimmune diseases such as MS. The ongoing phase II/III clinical trials are expected to further clarify E3s role and safety in treating MS patients. Furthermore, as suggested by the body of research reviewed, E3 appears to hold great promise as a potential therapy for numerous other autoimmune conditions yet to be explored.
E3 is currently available to practitioners through compounding pharmacies thus securing its appropriate place in the bio-identical hormone formulary.
AVAILABILITY OF E3 IN THE UNITED STATES
In recent years there has been an increase in demand and popularity of compounded bio-identical hormone replacement therapy (BHRT). This may be due, in part, to public awareness of side effects associated with prescription drugs such as Provera and others, and may also reflect increased consumer initiative in relation to their own health and well-being. E3 is available on prescription through a compounding pharmacy in the United States; however, there has been confusion with respect to its availability due to issuance of FDA letters to selected compounding pharmacies stating “no drug product containing E3 has been approved by the FDA and the safety and effectiveness of E3 is unknown.” In the letter, the FDA claimed the compounding of E3 was illegal and warned against claims of safety and efficacy. Also the FDA challenged the use of the term “bio-identical” claiming it has no accepted medical definition. In 2009, the FDA clarified its position stating that a ban was not imposed, rather, the FDA “respects a healthcare provider’s decision that his or her patient should receive estriol.”
The IACP responded to the political climate with rational facts and maintained the availability of E3. At present, compounding pharmacies provide E3 preparations and cite the following:
- In a press conference, the FDA admitted to not receiving a single report of an adverse event related to E3.
- E3 has a USP monograph. To compound E3 pursuant to a valid prescription is consistent with USP standards chapters 795 and 1075, Pharmacy Compounding Accreditation Boards’ standards and state pharmacy law.
- E3 has been used in Europe and Asia for many years. Its use is well supported in the medical literature. Loyd vs. Allen Jr., RPh reports that there are 39 approved E3 containing products in Europe and Asia, two produced by Wyeth: Cyclo-Menorette and Estriolsalbe (sold in Germany).
- The use of the term “bio-identical” describes precisely the nature of E3. It is human identical. FDA-registered manufacturers use the term on their websites to describe FDA-approved estrogen and progesterone products.
Furthermore, an E3 product is currently in phase III clinical trials at UCLA under the supervision of Dr. Rhonda Voskuhl as a therapeutic agent in the treatment of MS.
CONFLICT OF INTEREST STATEMENT
Dr Lommen and Dr Mead have formulated for and are minority shareholders in a company which has products that contain estriol; Kajarin Inc.
REFERENCES
- Lemon HM, Wotiz HH, Parsons L, et al. Reduced estriol excretion in patients with breast cancer prior to endocrine therapy.
- Head K. Estriol: safety and efficacy.
- Bergink EW. Oestriol receptor interactions: their biological importance and therapeutic implications.
- Lemon HM, Foley JF. Estrogens and risk of breast cancer.
- Lemon HM. Patholphysiologic consideration in the treatment of menopausal patients with oestrogens; the role of oestriol in the prevention of mammary carcinoma.
- Lemon HM. Experimental basis for multiple primary carcinogenesis by sex hormones: a review.
- Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial.
- Gruber CJ, Tschugguel W, Schneeberger C, Huber JC. Production and actions of estrogens.
- Nelson LR, Bulun SE. Estrogen production and action.
- Speroff L.
- Ganong WF.
- Lemon HM, Kumar PF, Peterson C, et al. Inhibition of radiogenic mammary carcinoma in rats by estriol or tamoxifen.
- Lemon HM, Parsons L. Reduced estriol excretion in patients with breast cancer prior to endocrine therapy.
- MacMahon B, Cole P, Brown JB, et al. Oestrogen profiles of Asian and North American women.
- Yang TS, Tsan SH, Chang SP, et al. Efficacy and safety of estriol replacement therapy for climacteric women.
- Takahashi K, Manabe A, Okada M, et al. Efficacy and safety of oral estriol for managing postmenopausal symptoms.
- Vooijs GP, Geurts TB. Review of the endometrial safety during intravaginal treatment with estriol.
- Cheury AC, Speck NM, de Moura KF, et al. Efficacy of vaginal use of topical estriol in postmenopausal women with urogenital atrophy.
- Kika G, Izumi S, Mori A, et al. Beneficial aspect of oral estriol as hormone replacement therapy: consideration on bone and lipid metabolism.
- Capobianco G, Donolo E, Borghero G, et al. Effects of intravaginal estriol and pelvic floor rehabilitation on urogenital aging in postmenopausal women.
- Soldan SS, Alvarez Retuerto AI, Sicotte NL, Voskuhl RR. Immune modulation in multiple sclerosis patients treated with the pregnancy hormone estriol.
- Voskuhl RR, Palaszynski K. Sex hormones in experimental autoimmune encephalomyelitis: Implications for multiple sclerosis.
- Sicotte NL, Liva SM, Klutch R, et al. Treatment of multiple sclerosis with the pregnancy hormone estriol.
- Damsker JM, Hansen AM, Caspi RR. Th1 and Th17 cells.
- Kim S, Liva SM, Dalal MA, et al. Estriol ameliorates autoimmune demyelinating disease: Implications for multiple sclerosis.
- Paperfuss TL, Powell ND, McClain MA, et al. Estriol generates tolerogenic dendritic cells in vivo that protect against autoimmunity.
- Zang YC, Halder JB, Hong J, Rivera VM, Zhang JZ. Regulatory effects of estriol on T cell migration and cytokine profile: Inhibition of transcription factor NF-κB.
- Lauritzen C. Results of a 5 year prospective study of estriol succinate treatment in patients with climacteric complaints.
- Palaszynski KM, Liu H, Loo KK, et al. Estriol treatment ameliorates disease in males with experimental autoimmune encephalomyelitis: Implications for multiple sclerosis.
- Gold SM, Voskuhl RR. Estrogen treatment in multiple sclerosis.
- Sribnick EA, Ray SK, Nowak MW, Li L, Banik NL. 17beta-estradiol attenuates glutamate-induced apoptosis and preserves electrophysiologic function in primary cortical neurons.
- Zhao L, Wu TW, Brinton RD. Estrogen receptor subtypes alpha and beta contribute to neuroprotection and increased Bcl-2 expression in primary hippocampal neurons.
- Pawlak J, Brito V, Kuppers E, Beyer C. Regulation of glutamate transporter GLAST and GLT-1 expression in astrocytes by estrogen.
- Liang Z, Valla J, Sefidvash-Hockley S, Rogers J, Li R. Effects of estrogen treatment on glutamate uptake in cultured human astrocytes derived from cortex of Alzheimer’s disease patients.
- Cantarella G, Risuglia N, Lombardo G, Lempereur L, Nicoletti F, Memo M, et al. Protective effects of estradiol on TRAIL-induced apoptosis in a human oligodendrocytic cell line: evidence for multiple sites of interactions.
- Sur P, Sribnick EA, Wingrave JM, Nowak MW, Ray SK, Banik NL. Estrogen attenuates oxidative stress-induced apoptosis in C6 glial cells.
- Dubal DB, Zhu H, Yu J, Rau SW, Shughrue PJ, Merchenthaler I, et al. Estrogen receptor alpha, not beta, is a critical link in estradiol-mediated protection against brain injury.
- Jover T, Tanaka H, Calderone A, Oguro K, Bennett MV, Etgen AM, et al. Estrogen protects against global ischemia-induced neuronal death and prevents activation of apoptotic signaling cascades in the hippocampal CA1.
- Murphy DD, Cole NB, Greenberger V, Segal M. Estradiol increases dendritic spine density by reducing GABA neurotransmission in hippocampal neurons.
- Rudick CN, Woolley CS. Estrogen regulates functional inhibition of hippocampal CA1 pyramidal cells in the adult female rat.
- Ziehn MO, Avedisian AA, Dervin SM, et al. Estriol preserves synaptic transmission in the hippocampus during autoimmune dymyelinating disease.
- Robinson DP, Klein SL. Pregnancy and pregnancyassociated hormones alter immune responses and disease pathogenesis.
- U.S. Department of Health and Human Services. U.S. Food and Drug Administration. FDA Takes Action Against Compounded Menopause Hormone Therapy Drugs. c2008. [cited 2012 Nov 14]. Available from:http://www.fda.gov/newsevents/newsroom/pressannouncements/2008/ucm116832.htm .
- http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/PharmacyCompounding/ucm183078.htm. Accessed July 31, 2013 .
- International Academy of Compounding Pharmacists. IACP Response to FDA Actions Against BHRT. c2008 [cited 2013 Feb 13]. Available from: http://iacprx.convio.net/site/DocServer/IACP_Letter_to_FDA.pdf?docID=3441 .
- Freedom Access to Natural Solutions. FDA, Wyeth, and Global Harmonization. c2008. [cited 2012 Dec 6